
International Journal of Optics and Photonic Engineering
(ISSN: 2631-5092)
Volume 4, Issue 2
Research Article
DOI: 10.35840/2631-5092/4521
Diabetes Chronicle Wound Analysis Using Terahertz Time Domain Spectroscopy and Imaging Technology
Ke Lin1*, Liu Hongwei2, Zhang Nan1, Wu Yang3, Sng Ming Keat4,5 and Tan Nguan Soon4,5,6,7
Table of Content
Figures
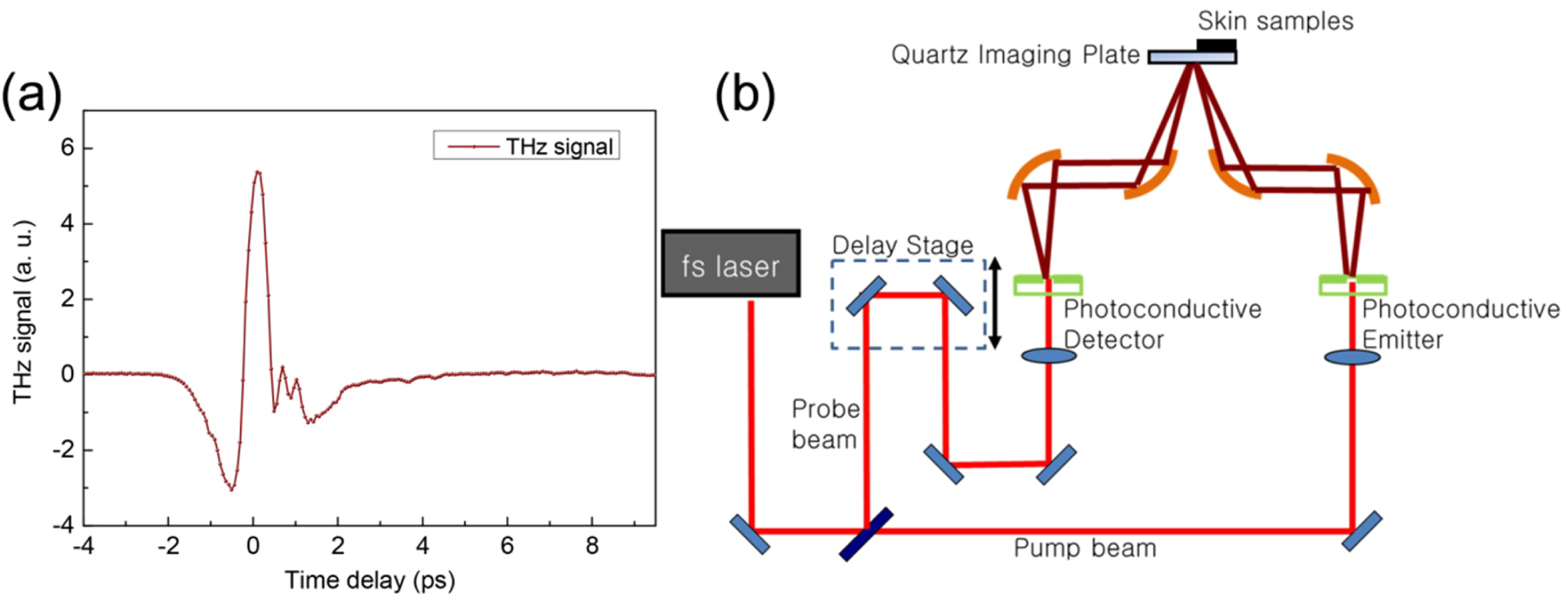
Figure 1: a) THz signal generated from the system...
a) THz signal generated from the system; b) Schematic Diagram of the solution of using THz for skin evaluation.
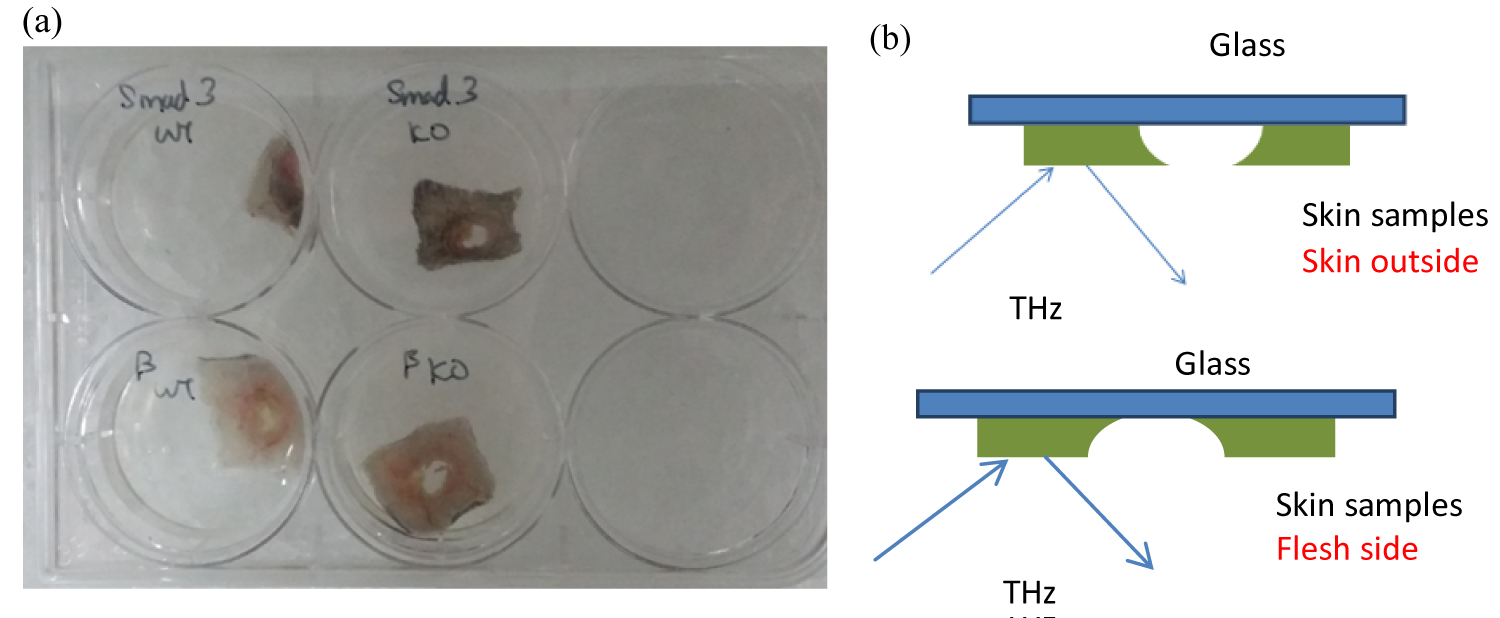
Figure 2: a) Samples under test; b) Diagram...
a) Samples under test; b) Diagram of the samples tested on the glass substrates.
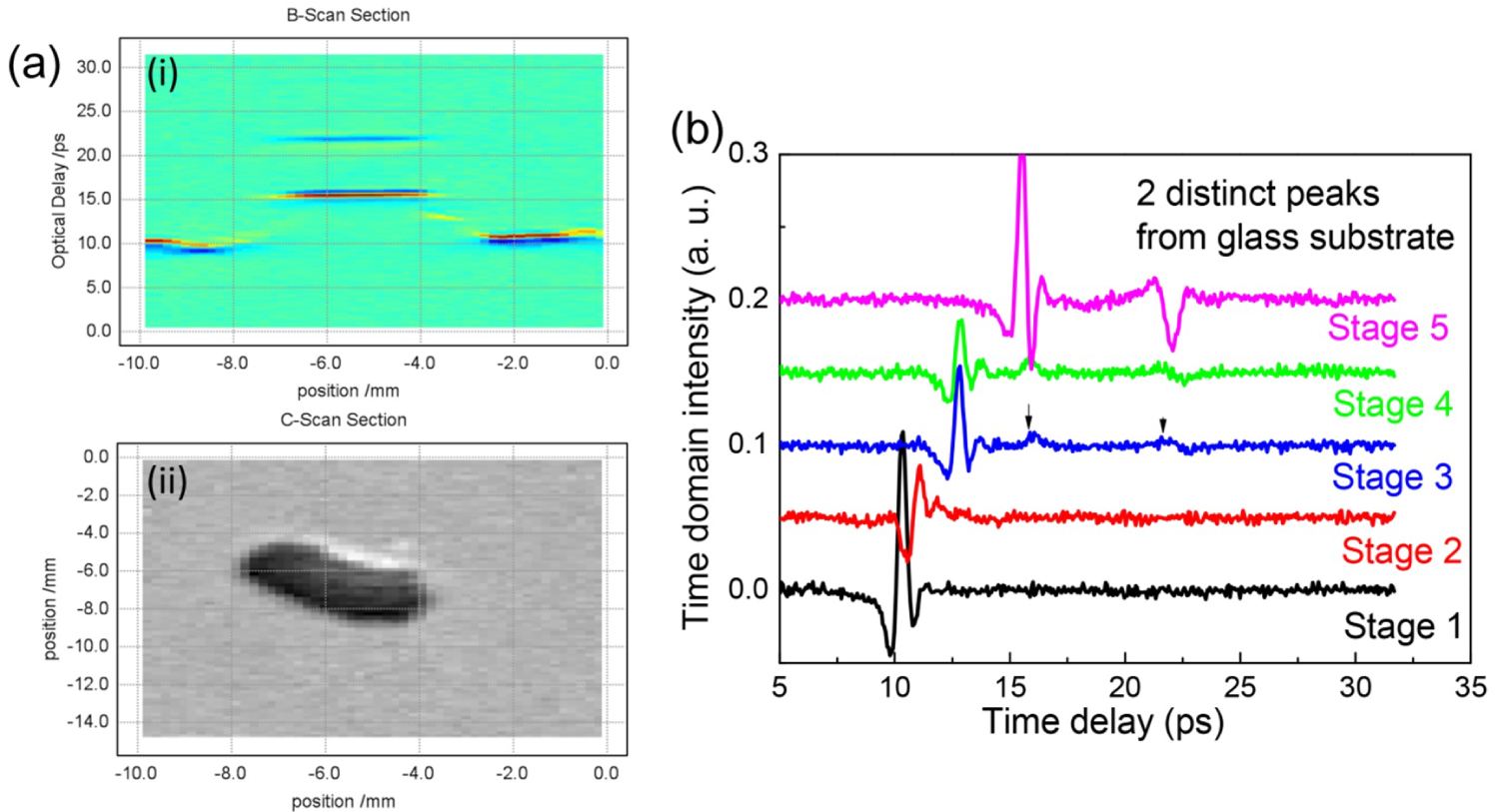
Figure 3: a) B-scan (i) and C-Scan (ii) of the wound...
a) B-scan (i) and C-Scan (ii) of the wound constructed based on the TDS signal; b) Series of curves scanned from good skin areas to the skin wound center.
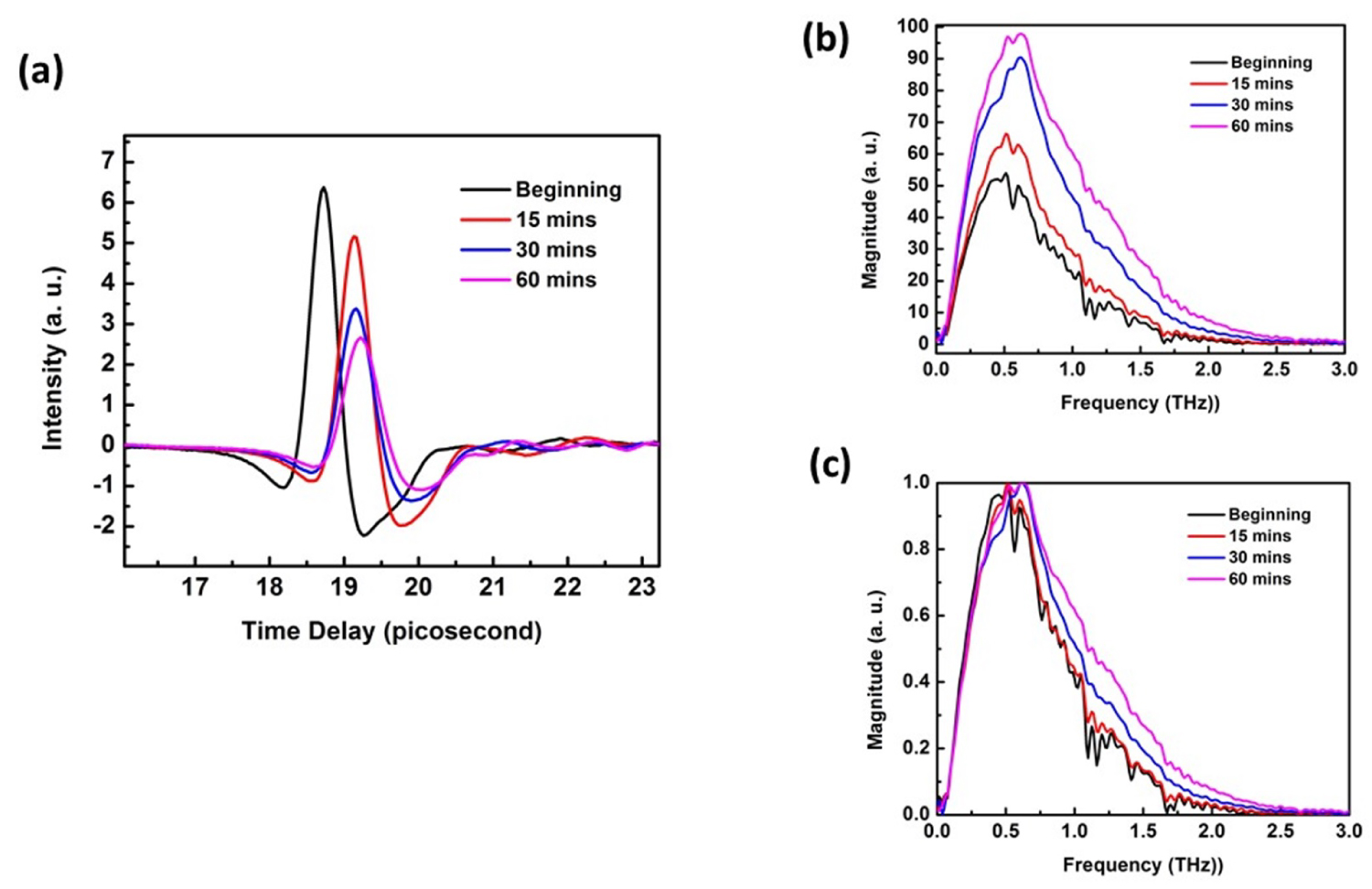
Figure 4: a) Time domain; b) Frequency domain spectral...
a) Time domain; b) Frequency domain spectral for skin at different hydration level; c) Normalized Frequency domain spectral for skin at different hydration level.
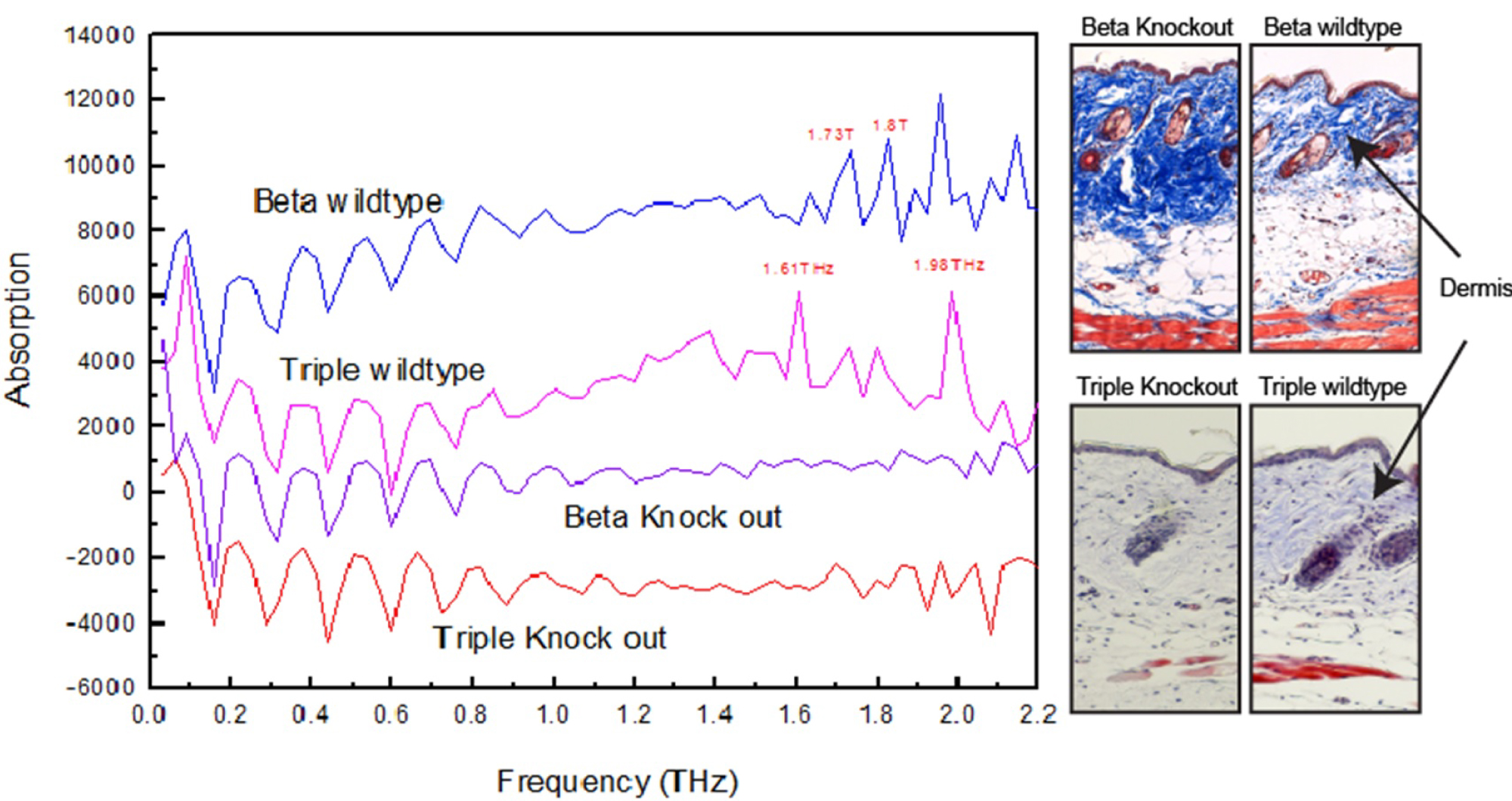
Figure 5: Absorption spectral calculated based on the...
Absorption spectral calculated based on the experimental data and staining histological pictures.
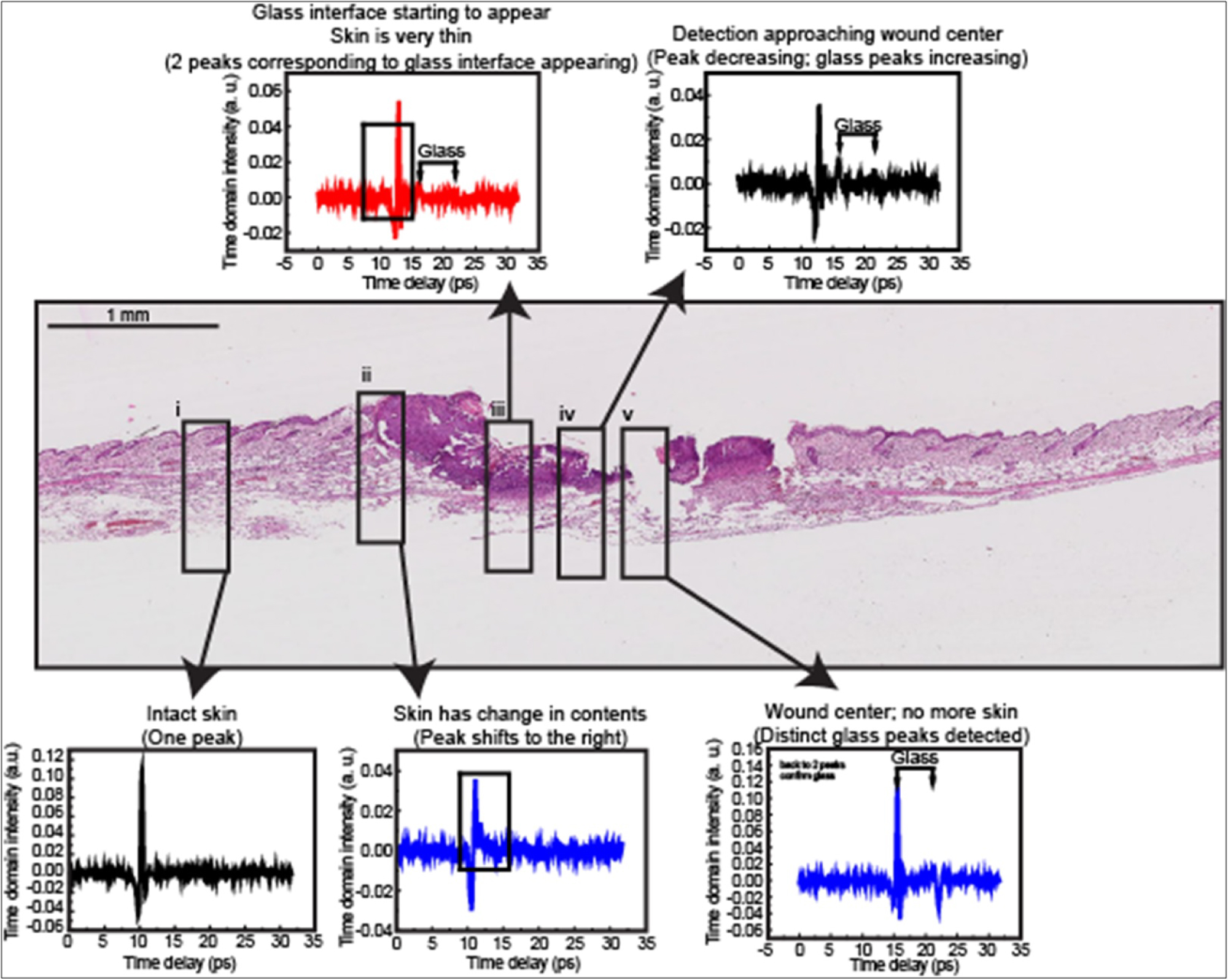
Figure 6: Skin wound B-scan pictures together...
Skin wound B-scan pictures together with the THz TDS probe swept from the left of the good skin area towards the centre of the skin wound.
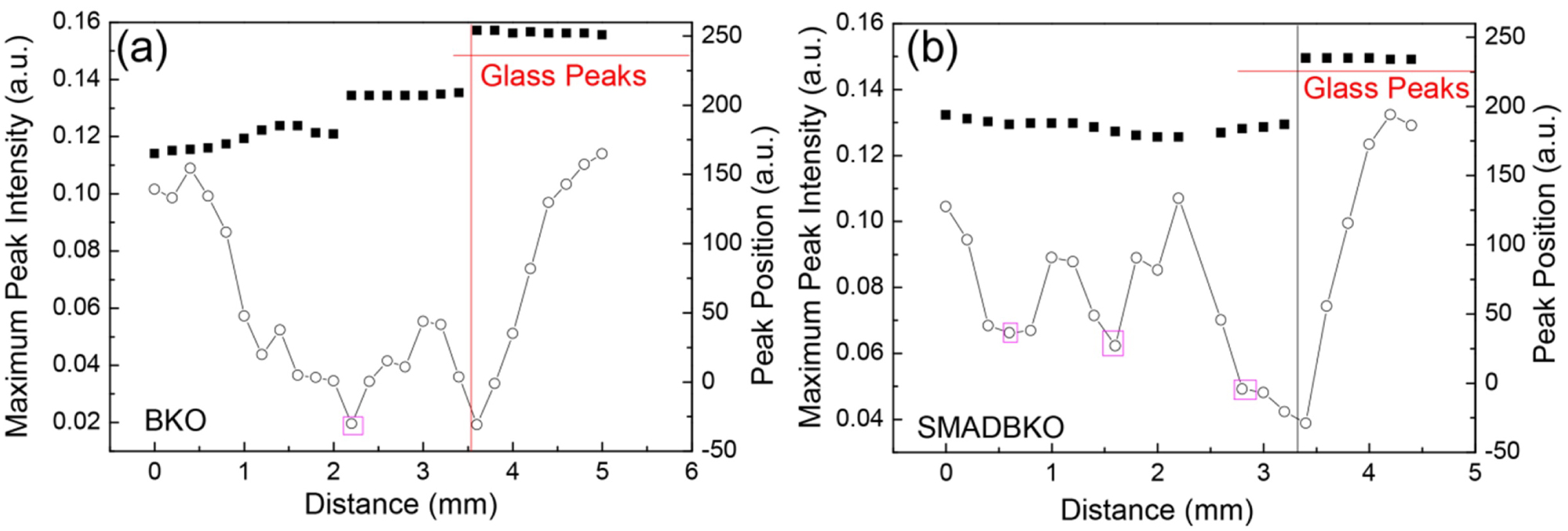
Figure 7: TDS peak intensity and peak position...
TDS peak intensity and peak position changes versus the distance to the wound center for a) Beta Knock Out sample and b) Triple Knock Out sample.
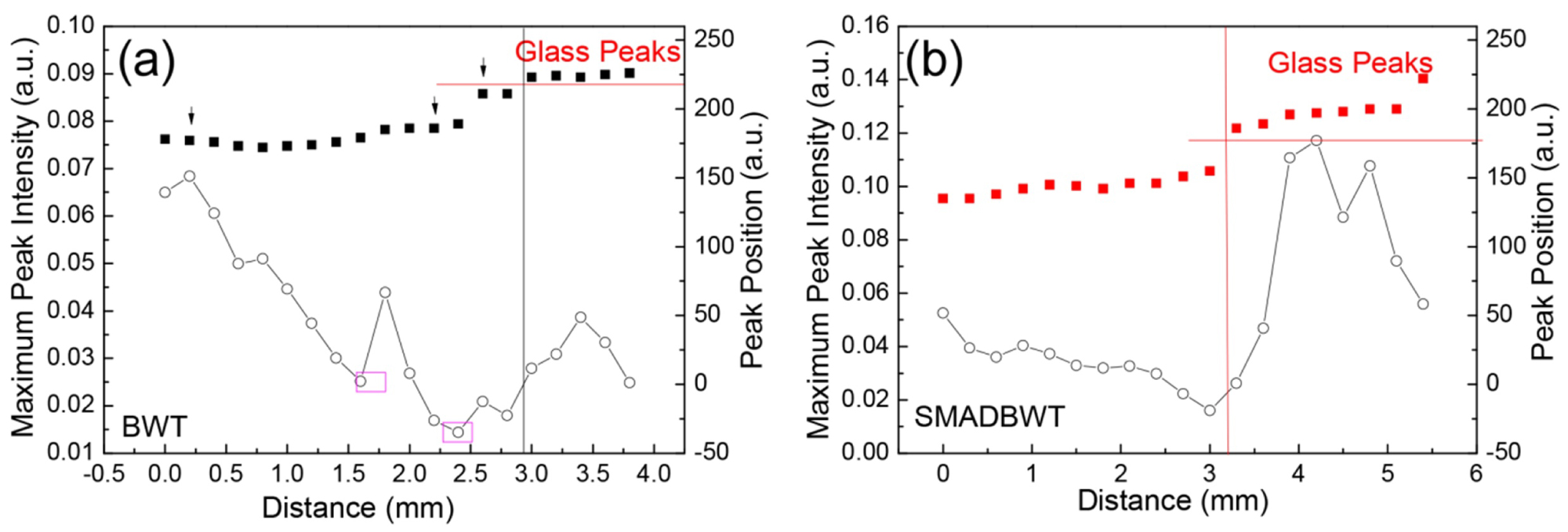
Figure 8: a) TDS peak intensity and peak position...
a) TDS peak intensity and peak position changes versus the distance to the wound center for a) Beta Wild type samples; b) Triple Wild type.
References
- Chua TG, Goh CL, Koh SL (1992) Pattern of skin diseases at the National Skin Center (Singapore ) From 1989-1990. Int J Dermatol 31: 555-559.
- Ho SGY, Chan HHL (2009) The Asian dermatologic patient: review of common pigmentary disorders and cutaneous diseases. Am J Clin Dermatol 10: 153-168.
- Sng J, Koh D, Siong WC, Choo TB (2009) Skin cancer trends among Asians living in Singapore from 1968 to 2006. J Am Acad Dermatol 61: 426-432.
- Ferguson MWJ, Whitby DJ, Shah M, Armstrong J, Siebert JW, et al. (1996) Scar formation: The spectral nature of fetal and adult wound repair. Plast Reconstr Surg 97: 854-860.
- Ferguson MWJ, O'Kane S (2004) Scar-free healing: From embryonic mechanisms to adult therapeutic intervention. Philos Trans R Soc Lond B Biol Sci 359: 839-850.
- Harty M, Neff AW, King MW, Mescher AL (2003) Regeneration or scarring: An immunologic perspective. Dev Dyn 226: 268-279.
- Junker JPE, Philip J, Kiwanuka E, Hackl F, Caterson EJ, et al. (2014) Assessing quality of healing in skin: Review of available methods and devices. Wound Repair Regen 22: 2-10.
- Pickwell E, Wallace VP (2006) Biomedical applications of terahertz technology. J Phys D: Appl Phys 39.
- Yang X, Zhao X, Yang K, Liu Y, Liu Y, et al. (2016) Biomedical applications of terahertz spectroscopy and imaging. Trends Biotechnol 34: 810-824.
- Alhuwaidi S, Zubair K, Song H, Shellman Y, Robinson W, et al. (2015) Disease diagnostics of biological tissues using free-space technique in terahertz frequency range. Biomedical Circuits and Systems Conference (BioCAS), IEEE, Atlanta, GA, USA.
- Mantsch HH, Naumann D (2010) Terahertz spectroscopy: The renaissance of far infrared spectroscopy. J Mol Struct 964: 1-4.
- Wu H, Heilweil EJ, Hussain AS, Khan MA (2007) Process analytical technology (PAT): Effects of instrumental and compositional variables on terahertz spectral data quality to characterize pharmaceutical materials and tablets. Int J Pharm 343: 148-158.
- Cherkasova OP, Nazarovb MM, Berlovskayac EE, Angelutsc AA, Makurenkovc AM, et al. (2016) Studying human and animal skin optical properties via terahertz pulsed spectroscopy. Bulletin of the Russian Academy of Sciences Physics 80: 479-483.
- Hernandez-Cardoso GG, Rojas-Landeros SC, Alfaro-Gomez M, Hernandez-Serrano AI, Salas-Gutierrez I, et al. (2017) Terahertz imaging for early screening of diabetic foot syndrome: A proof of concept. Scientific Reports 7: 42124.
- Son JH (2009) Terahertz electromagnetic interactions with biological matter and their applications. J Appl Phys 105: 102033.
- Kim KW, Kim KS, Kim H, Lee SH, Park JH, et al. (2012) Terahertz dynamic imaging of skin drug absorption. Opt Express 9: 9476-9484.
- Exter M, Fattinger C, Grischkowsky D (1989) Terahertz time-domain spectroscopy of water vapor. Opt Lett 20: 1128-1130.
- Ortolani M, Lee JS, Schade U, Hübers HW (2008) Surface roughness effects on the terahertz reflectance of pure explosive materials. Appl Phys Lett 93: 081906.
- Withayachumnankul W, Fischer BM, Abbott D (2008) Numerical removal of water vapor effects from terahertz time-domain spectroscopy measurements. Proc R Soc A 464: 2435-2456.
- Kemp M C (2011) Explosives detection by terahertz spectroscopy-A bridge too far? IEEE Trans Terahertz Sci Technol 1: 282-292.
- Jepsen PU, Fischer BM (2005) Dynamic range in terahertz time-domain transmission and reflection spectroscopy. Opt Lett 30: 29-31.
- Siegel P (2004) Terahertz technology in biology and medicine. IEEE Transactions on Microwave Theory and Techniques 52: 2438-2447.
- Macpherson E, Gallerano GP, Park GS, Hintzche H, Wilmink GJ (2013) Guest editorial: Terahertz imaging and spectroscopy for biology and biomedicine. IEEE Trans on Terahertz Science and Technology 3: 354-356.
- Arnone D, Ciesla C, Pepper M (2000) Terahertz imaging comes into view. Physics World 4: 35-40.
- Crawley D, Longbottom C, Wallace VP, Cole B, Arnone D, et al. (2003) Three-dimensional terahertz pulse imaging of dental tissue. J Biomed Opt 8: 303-307.
- Woodward RM, Wallace VP, Arnone D, Linfield EH, Pepper M (2003) Terahertz pulsed imaging of skin cancer in the time and frequency domain. J Biol Phys 29: 257-259.
- Pickwell E, Cole BE, Fitzgerald AJ, Pepper M, Wallace VP (2004) In vivo study of human skin using pulsed terahertz radiation. Phys Med Biol 49: 1595-1607.
- T Ikeda, A Matsushita, M Tatsuno, Y Minami, M Yamaguchi, et al. (2005) Investigation of inflammable liquids by terahertz spectroscopy. Appl Phys Lett 87: 034105.
Author Details
Ke Lin1*, Liu Hongwei2, Zhang Nan1, Wu Yang3, Sng Ming Keat4,5 and Tan Nguan Soon4,5,6,7
1Institute of Materials Research and Engineering, A*STAR Research Entitles, 2 Fusionopolis Way, Innovis, #08-03, Singapore
2Jiangsu Key Lab on Opto-electronic Technology, School of Physics and Technology, Nanjing Normal University, China
3Department of Electrical & Computer Engineering, Faculty of Engineering, National University of Singapore, Singapore
4School of Biological Sciences, Nanyang Technological University, Singapore
5Lee Kong Chian School of Medicine, Nanyang Technological University, Singapore
6Institute of Molecular and Cell Biology, 61 Biopolis Drive, Proteos, Singapore
7KK Research Centre, KK Women's and Children's Hospital, Singapore
Corresponding author
Ke Lin, Institute of Materials Research and Engineering, A*STAR Research Entitles, 2 Fusionopolis Way, Innovis, #08-03, 138634, Singapore, Tel: +65-63194913, Fax: +65-8727528.
Accepted: December 18, 2019 | Published Online: December 20, 2019
Citation: Lin K, Hongwei L, Nan Z, Yang W, Keat SM, et al. (2019) Diabetes Chronicle Wound Analysis Using Terahertz Time Domain Spectroscopy and Imaging Technology. Int J Opt Photonic Eng 4:021
Copyright: © 2019 Lin K, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
The terahertz (THz) wave technology was demonstrated for investigating skin wound healing process based on our in house constructed THz system. The time-domain signals data were collected, B-scan and C-scan images were reconstructed from the untouched skin part to the wound center continuously. The hydration level, absorption characteristic peaks of the skin compositions can be identified and correlated to the histologically stained imaging results. The direct and non-invasive access to the information of skin compositions would allow for the further study of the chronic wounds formation mechanism as well as the reasons for the dysregulation process happened in diabetic patients. Thereafter drug development and precise targeted treatment can be further studied.
Keywords
Portable THz system, Skin, Wound healing
Introduction
Poor healing and chronic wounds are those that have failed to proceed through a tightly and orderly regulated reparative process, resulting in a compromised anatomic and functional integrity of the injured site [1-3]. One of the most debilitating complications in diabetes is refractory wound healing, resulting in severe complications that frequently cause morbidity and mortality [4-6]. With only one FDA-approved cytokine-based treatment available to manage diabetic ulcers, there is an urgent need to devise better methods to elucidate differences between normal and diabetic wounds, such that novel drugs that target these components precisely can be designed [7]. However, researchers face the difficulty of obtaining biopsies from diabetic patients, which may lead to undesired consequences. Therefore, it is important to devise a non-invasive method to study patients' wounds. With this growing demand, it is imperative that dermatologists provide not only preventative counseling but also be astute in the early diagnosis and treatment of any suspected malignancies. Emerging diagnosis tools are non-invasive imaging devices that allow for the visualization of skin without the traditional excisional biopsy. These tools have many promising features which are not available through routine histology: Examination of the whole lesion rather than a section of the lesion, temporal management of lesions, with the ability to track their growth and change by reviewing past images, as well as monitoring the effectiveness of topical treatments by examining changes in cellular structure over time. While the capabilities of non-invasive imaging tools are far-reaching, each tool varies in its resolution depth, clarity of image, applicability in a clinical setting, accuracy, sensitivity, and specificity.
The THz wave technology has demonstrated great potential for medical diagnostic applications [8-10] as it is not ionizing radiation and its low photon energies do not damage tissues and DNA. The potential applications are expanded even further with the increased availability of many absorption spectra (i.e., fingerprint spectra) particular to specific molecular structures. THz time-domain spectroscopy (THz-TDS) has been demonstrated as an excellent tool for the noninvasive assessment of skin and tissues measurement [11-16]. Despite the far-reaching potential of terahertz spectroscopy, there are still limitations [17-20] to the use of THz technology, such as atmospheric absorption, especially due to water vapor [17]. The limited penetration depth also hinders the technology applications in medical applications. The limitations are largely due to the low THz source power. A more compact system with higher power, higher dynamic range, higher signal/noise ratio, lower price, less maintenance and ideally a “push-button” level of complexity are facing worldwide THz researchers. In our work, we developed an accurate in vivo instant measurement system to extract skin compositions distribution such as: Hydration, keratinocytes, fibroblasts etc. The skin composition differences between normal and diabetic wound can be reviewed. The direct, yet non-invasive, access to the information of skin composition would allow for further studies of the chronic wounds formation mechanism, as well as the better understanding of the reasons for the dysregulation process occurring in diabetic patients. Thereafter drug development and precise targeted treatment can be further studied. The compact THz spectrometer permits time domain in-situ measurement of skin, collection of the data and calculation of the absorption spectrum and optical properties that were comparable to those collected with conventional commercial systems. The goal of our study was to study the different types of diabetic skin healing process using compact THz system by measuring the optical parameters of the subjects. Spectral were collected from 0.1 to 3 THz. The results for this study will provide a framework for using compact THz spectrometers for clinical applications.
Experimental Section
Instrumentation
A compact THz spectrometer was developed in our lab. Figure 1a shows the THz signal generated from our in-house built THz system based on a DC motor delay line. It shows good signal to noise level. Figure 1b is the schematic diagram of the TDS system using for skin evaluation. The system includes: THz wave generation and detection unit. The unit hardware incorporated compact robust femtosecond laser head and a fast mechanical-free optical delay line developed in-house in Institute of Materials Research and Engineering, Agency for Science, Technology and Research (IMRE, A*STAR), which can obtain THz spectrum with high speeds and signal to noise ratio. The advantages of the all-fiber-coupled design and mechanical-free optical delay line include improved portability and minimal maintenance, in contrast to the current bulky and fragile THz systems in the labs. The compact THz system is less than 40 cm (W) × 40 cm (L) × 15 cm (H) and the weight is less than 15 kg. The software algorithm calculate the absorption coefficient and other optical parameters, translates the time domain THz pulse signal data into information related to the skin pathology, such as: The concentrations of skin tissue components, e.g. keratinocytes and fibroblasts.
Theoretically understanding the skin composition behaviors under THz spectroscopy
In the THz-TDS, the form of the electric field is shaped as a THz pulse. The THz pulse can be detected after the interactions with the sample material as a function of the time delay. Differences of pulse amplitude maximum and minimum, integral pulse value of a certain previously fixed interval on the time delay scale or the relative time delay shift in relation to a reference time point are considered to reveal sample's specific information. More detailed spectral information on the optical properties of the samples can be obtained if the THz pulse in the time domain is done with Fast Fourier Transformed (FFT). The resulting frequency-dependent amplitude and phase spectrum can distinguish the materials and facilitate the estimation of the complex refraction index or the dielectric permittivity, respectively. Thereafter, in our application, the skin components with unique fingerprints can be analyzed based on the THz frequency spectral arising from their intermolecular vibrations and the intramolecular mode.
In reflection configuration under a normal incidence, the incoming THz wave is reflected at the surface of the tested sample without propagating through the absorbing material. In the frequency domain the ratio between the sample and the reference spectra is then [21]
Above equation can be inverted, and the simple expression for the index of refractive and absroption coefficient can be found:
With this system, skin clinicians will be able to better provide targeted and customized treatments, as the instant components of the skin tissue information will reveal the spectral differences between a normal and diabetic wound.
Absorption spectral peak positions in THz range according to different compositions have to be simulated and identified. Drug absorption peaks shall be identified and monitored. The models for extraction of the skin compositions have to be setup.
Sample preparation
Experimental mice were utilized in this work. Samples were supplied from the Animal Research Facility, Nanyang Technological University. The facility runs on a 12 h/12 h light/dark cycle, and animals were provided with chow and water ad libitum. To harvest skin biopsies for histological staining, we anaesthetized the animal before obtaining skin biopsies from the dorsal backs. Harvested biopsies were washed in ice-cold 1X phosphate-buffered saline (PBS) before being fixed in 4% paraformaldehyde. Fixed tissues were subsequently be dehydrated over an ethanol gradient and xylene before being embedded in paraffin wax. Embedded tissues were sectioned at a thickness of 5 mm. Sectioned tissues were rehydrated over an ethanol gradient in the reverse direction before being stained for necessary histological studies. Animal experiments were carried out in accordance to the guidelines of the University Institutional Animal Care and Use Committee (IACUC, ARF-SBS/NIE-A0216AZ, A0321, A0322), Singapore.
All TDS measurement were carried on under N2 gas environment, so the humidity effect has been removed.
Results and Discussion
TDS image analysis
Figure 2a shows the outlook of the samples under test. Samples either show scar or scar less structures. Figure 2b shows the diagram of the samples tested on the glass substrates. THz-TDS has been used to detect from both the skin side and inner flesh side. The following analysis results are based on the test from skin outside.
Figure 3a shows cross section scan (B-scan) and surface scan (C-Scan) of the wound constructed based on the TDS signal. TDS spectral was collected from the unwounded portion of the skin with steps 200 um towards the wound center. The TDS spectra displayed only one reflection peak from the skin surface. With the monitoring moving towards the wound center, the TDS peak shifted and a new peak from the bottom interface with glass substrate appeared. At the center of the wound, two clear peaks from the top and bottom of the glass substrate reflection peaks appeared. Figure 3b shows the series of curves together, and Y-axis was adjusted for a better comparison. Curves 1-5 corresponded to the scans from the unwounded portion of the skin to the wound center, which also corresponded to the five stages of skin changes from good skin, thinner skin, healing skin, scar formation stage and damaged skin center. The measurement goes through from stage 1 to stage 5 for all the samples. At stage 1, the THz spectral displayed a distinct peak from the surface of the good skin. From stages 2 to 4, the skin peak started to decrease and shifted to the right, indicating either a thinning of the skin or a production of new elements. Progressively, the peaks from the glass substrate top and bottom interfaces began to appear. As the skin became thinner, the THz peak from skin part decreased and the THz reflection peaks from glass interfaces became more obvious. At stage 5, skin THz reflection peaks disappeared completely, with the concurrent appearances of two distinct THz reflection peaks from glass interfaces, indicating the total absence of skin tissue.
Skin hydration level comparison
THz radiation has lots of advantages in medicine and medical applications [22-27]. It is a nondestructive method and won't damage the samples. In this application, we imaged the changes in skin samples from which were taken out from saline water to different dehydrated time period, in order to investigate the skin's hydration level. Figure 4a is the time-domain signal for the moisturized skin and the skin dehydrated at 15 mins, 30 mins and 60 mins. Figure 4b and Figure 4c are the frequency-domain spectral for the moisturized skin and the skin dehydrated at 15mins, 30 mins and 60 mins. Figure 4c is the normalized results from Figure 4b.
From Figure 4a, we can see that when the samples were left in air to dehydrate, the signal intensity decreases with the dehydration time increases. The THz reflection signal intensity of sample at dehydrated state is much lower than that of moisturized state. In addition, delay time increased compared with the moisturized skin. When the sample dehydrated, the THz wave was able to penetrate into the tissue structure. Thus the optical path length increased accordingly. Figure 4b demonstrated that the reflection intensity increased with the dehydration time increased which means the water absorption for THz frequency range were reduced. With the longer dehydration time, more water lost from the skin surface, water absorption in THz range continuous decreased. When we normalize the frequency domain spectral, as shown in Figure 4c, the intensity increases with dehydration time increases is more obvious in the range between 0.8 THz and 1.7 THz in which most of the water absorption happened [28]. The THz-TDS is suitable for the characterization of hydration level of the skin by application of the corresponding reflection mode.
Skin wound formation analysis
Investigations performed on whole mount intact skin tissues revealed that the THz detection could detect differences in content of skin tissues. Skin biopsies from animal models with dermal fibrosis (indicated as Beta Knockout) were studied alongside their respective wild type littermates. We performed Masson's Trichrome staining on the tissues. The left column in Figure 5 shows the absorption spectral calculated based on Equation (1) and (2) which were discussed in experimental section. The right column in Figure 5 shows the staining histological pictures. In this histological stain, the intensity of blue color indicates the amount of collagen deposition in the dermis. Histologically, we were able to observe that Beta Knockout had thicker dermis and more collagen content than the Beta wild type. This change in dermal content was demonstrated in our THz spectra, in which the peaks at 1.73 THz and 1.8 THz disappeared in the Beta Knockout tissue as shown in Figure 5 left column.
Using this as a positive control, we looked at skin tissues from Triple Knockout mice. Although Masson's Trichrome staining revealed that Triple knockout had slightly lesser collagen deposition, thickness of the dermis between Triple Knockout and Triple wild type had no significant differences. This could be due to the presence of other contents which we were unable to discern visually through histology analyses. However, our THz spectra was able to detect that there were differences in content between the Triple Knockout and Triple wild type, based on the disappearance of peaks at 1.61 THz and 1.98 THz. Although the Triple Knockout did not exhibit a thicker dermis like the Beta Knockout did, content changes could be due to other reasons such as changes in lipid content.
Figure 6 shows skin wound B-scan pictures together with the THz-TDS probe swept from the unwounded skin area towards the center of the skin wound. The probe detected peak shifts that occurred in accordance to the contents within the wounded tissue itself. From Figure 6, it can be observed: (i) The probe detected a single peak corresponding to the unwounded section of the skin tissue; (ii) As the probe moved towards the wound center, the probe detected a change in tissue composition at the section nearer to the wound center where a massive proliferation of cells, such as fibroblasts, was occurring to support the wound closure process; (iii) As the probe entered the region of the wound bed, the tissue became thinner as regeneration has not occurred in this area yet and the probe detected some portions of the glass interface; (iv) Increasing surface area of the glass interface began to occur as probe approached wound center; and (v) Probe eventually reached the center of the wound where tissue is minimal, with maximum exposure of the glass interface to the probe.
Scar behavior observation during healing process
TDS peak intensity and peak positions along the scanning distance from the unwounded section of the skin to the wound center have been plotted. Figure 7a shows the TDS peak intensity and peak position changes versus the distance to the wound center for Beta Knockout sample. Figure 7b shows the TDS peak intensity and peak position changes versus the distance to the wound center for Triple Knockout sample. For both Knockout samples, the curves showed distinct intensity peaks, which could reveal different structures or tissues being generated during wound healing, for example, scar formation. We believe these generated tissue could hinder the wound healing process. Compared Figure 7a and Figure 7b, the Beta Knockout sample has a higher jump than the Triple Knockout sample at the 2.5 mm, which means the tissues being generated in Beta Knockout has more scar characterization than Triple Knockout sample.
Figure 8a and Figure 8b show the TDS peak intensity and peak position changes versus the distance to the wound center for Beta wild type samples and Triple wild type samples. Both samples has a similar behavior. There is no clear peak position jump with the distance variation, which means the scar tissue is not obvious. Compared to the Knockout samples with “sharp” peaks, both wild type samples have only small surges, the surge is further away from the wound center and much smoother. The healing of wild type sample looks better than Knockout type sample.
Thus, through this measurement and analysis, the scar behavior during the healing process and be observed.
Conclusion
Our approach is novel in the way that it uses a THz pulse to detect different skin tissues and interfaces, through FFT to obtain frequency spectral, absorption spectral can be derived. The hydration level and skin composition characteristic peaks can be identified and extracted. There is no related report regarding this research work in the said field. Through the analysis of good skin and diabetic skin healing process using non-invasive THz technology, the series of events including inflammation, re-epithelialization and matrix remodeling can be analyzed. The results will help to understand the reasons of the dysregulation of these physiological events in the case of chronic wounds in diabetic patients. Our approach is technologically significant in the way that it allows one to obtain the wound healing composition information during different stages. Our prototype can obtain the measurement in milli-seconds which will cause no disruption, no damage and no pain for the patients. Through investigating the skin compositions during healing, the drug effects for wound healing can also be analyzed.

