International Journal of Metallurgy and Metal Physics
(ISSN: 2631-5076)
Volume 4, Issue 1
Research Article
DOI: 10.35840/2631-5076/9231
Preparation and Performance of Nitrogen-containing Stainless Steel
Dong Li1, Jiqiang Wu1,2, Shijing Lu1, Bin Miao1, Wei Cai1 and Jing Hu1,2*
Table of Content
Figures
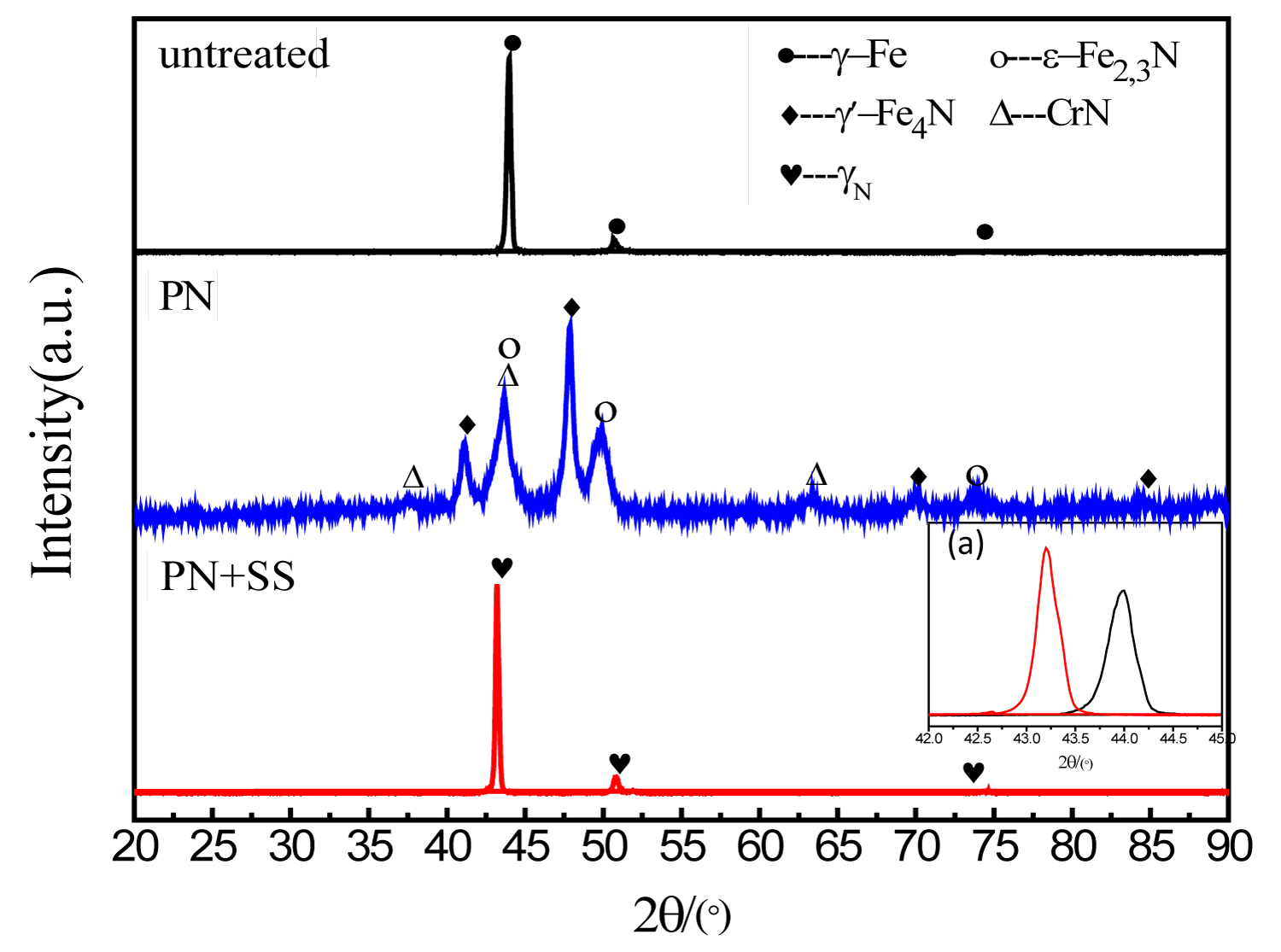
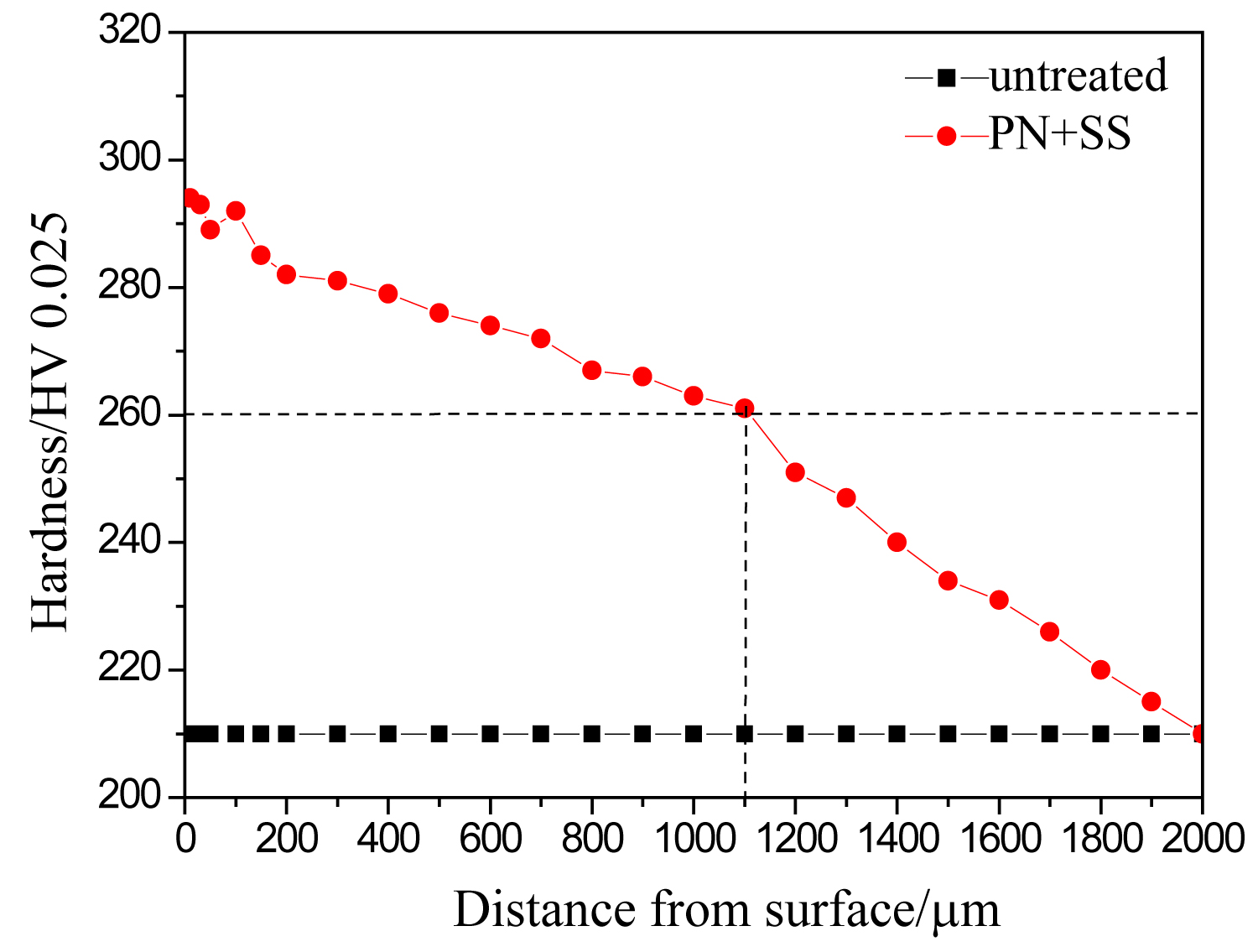
Figure 2: Micro-hardness profile of samples...
Micro-hardness profile of samples untreated and treated by the complex process.
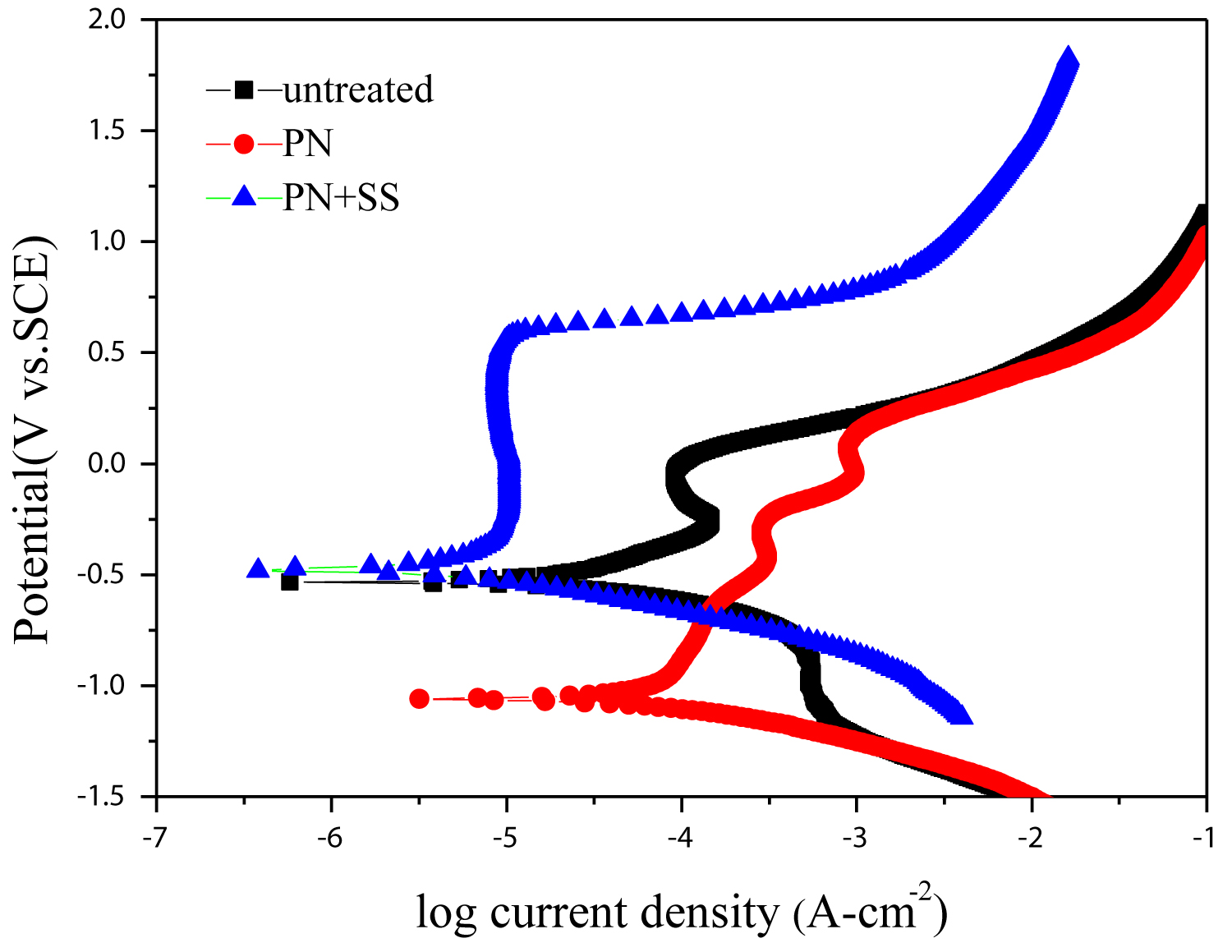
Figure 3: Potentiodynamic polarization curves...
Potentiodynamic polarization curves of samples treated by different processes.
References
- Wu JQ, Liu H, Ye XM, Chai YT, Hu J (2015) Enhancement of corrosion resistance for plasma nitrided AISI 4140 steel by plain air plasma post-oxidizing. J Alloys Compd 632: 397-401.
- Miao B, Song L, Chai YT, Wei KX, Hu J (2017) The effect of sand blasting pretreatment on plasma nitriding. Vacuum 136: 46-50.
- Simmons JW (1996) Overview: High-nitrogen alloying of stainless steel. Mater Sci Eng A 207: 159-165.
- Menthe E, Bulak A, Olfe J, Zimmermanna A, Riea KT (2000) Improvement of the mechanical properties of austenitic stainless steel after plasma nitriding. Surf Coat Technol 133: 259-263.
- Wang KS, Che HL, Lei MK (2016) Corrosion-fatigue properties of plasma-based low-energy nitrogen ion implanted AISI 304 L austenitic stainless steel in borate buffer solution. Surf Coat Technol 288: 30-35.
- Li Y, Zhang SZ, He YY, Zhang L, Wang L (2014) Characteristics of the nitrided layer formed on AISI 304 austenitic stainless steel by high temperature nitriding assisted hollow cathode discharge. Mater Design 64: 527-534.
- Wu J Q, Liu H, Li JC, Yang XM, Hu J (2016) Comparative study of plasma oxynitriding and plasma nitriding for AISI 4140 steel. J Alloy Compd 680: 642-645.
- Masayuki A, Yoshihiko U, Toshifumi K, Nakajima M, Tsuchiyama T, et al. (2015) Effect of sensitization on corrosion fatigue behavior of type 304 stainless steel annealed in nitrogen gas. Mater Sci Eng A 640: 33-41.
- Martinavicius A, Abrasonis G, Scheinost AC, Danoix R, Danoix F, et al. (2012) Nitrogen interstitial diffusion induced decomposition in AISI 304L austenitic stainless steel. Acta Mater 60: 4065-4076.
- Lin Y, Lu J, Wang L, Xu T, Xue Q (2006) Surface nanocrystallization by surface mechanical attrition treatment and its effect on structure and properties of plasma nitrided AISI 321 stainless steel. Acta Mater 54: 5599-5605.
- Pan TJ, Chen Y, Zhang B, Hu J, Li C (2016) Corrosion behavior of niobium coated 304 stainless steel in acid solution. Appl Surf Sci 369: 320-325.
- Li Y, Wang Z, Wang L (2014) Surface properties of nitrided layer on AISI 316L austenitic stainless steel produced by high temperature plasma nitriding in short time. Appl Surf Sci 298: 243-250.
- Li JC, Yang XM, Wang SK, Wei KX, Hu J (2014) A rapid D. C. plasma nitriding technology catalyzed by pre-oxidation for AISI4140 steel. Mater Lett 116: 199-202.
- Fewell MP, Priest JM, Baldwin MJ, Collins GA, Short KT (2000) Nitriding at low temperature. Surf Coat Tech 131: 284-290.
Author Details
Dong Li1, Jiqiang Wu1,2, Shijing Lu1, Bin Miao1, Wei Cai1 and Jing Hu1,2*
1Jiangsu Key Laboratory of Materials Surface Science and Technology, Changzhou University, China
2Sinopec Oilfield Equipment Corporation, Kingdream Public Limited Company, China
Corresponding author
Jing Hu, Jiangsu Key Laboratory of Materials Surface Science and Technology, Changzhou University, Changzhou, 213164; Sinopec Oilfield Equipment Corporation, Kingdream Public Limited Company, Wuhan, 430223, China, Tel: 86-0519-86330095.
Accepted: May 16, 2019 | Published Online: May 18, 2019
Citation: Li D, Wu J, Lu S, Miao B, Cai W, et al. (2019) Preparation and Performance of Nitrogen-containing Stainless Steel. Int J Metall Met Phys 4:031.
Copyright: © 2019 Li D, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Nitrogen-containing stainless steel was prepared by a novel combination of plasma nitriding and solid solution. The performance of the treated samples was evaluated by X-ray diffraction (XRD), micro-hardness test and electrochemical polarization. The results showed that a thick nitrogen-containing layer composed of nitrogen expanded austenite (γN) layer was obtained by the novel technology, and the corresponding γN peaks appeared at lower angles comparing with those of the untreated sample. Meanwhile, both the microhardness and corrosion resistance were enhanced in comparison with those of the untreated sample.
Keywords
Nitrogen-containing, Austenitic stainless steel, Plasma nitriding, Solid solution treatment
Introduction
As was reported that the hardness and strength of austenitic stainless steel can be significantly improved with the addition nitrogen and the corrosion resistance can be further enhanced as well [1-4]. The existed methods to produce nitrogen-containing austenitic steel include high pressure melting and high temperature gas nitriding etc. [5-8]. Unfortunately, these methods are hard to be carried out in practical applications; especially they are restricted by the demand of special equipments. Therefore, it is of significant value to develop a new method to prepare nitrogen alloyed stainless steels.
Plasma nitriding is a kind of chemical heat treatment technology, during which the nitrogen atoms can diffuse into the metals by ion sputtering to form nitriding layer with much higher nitriding efficiency comparing with most other existing nitriding technologies [9-11]. Unfortunately, plasma nitriding of austenitic stainless steel is usually accompanied with the precipitation of chromium nitrides, which brings out the deterioration of the corrosion resistance due to the depletion of the chromium content in the matrix. Though a single S-phase without chromium nitrides precipitation in the nitriding layer can be formed at temperatures lower than 450 ℃, the rate of plasma nitriding process is very slow at this low temperature, leading to very long duration for getting deep nitriding layer, which results in very low production efficiency and high cost in real application [12,13].
Solid solution is a kind of heat treatment by heating the treated metal to high temperature and holding for enough time for fully decomposing the compounds existed in the matrix, and then rapidly cooling to get a supersaturated solid solution [14].
Based on the characteristics of both plasma nitriding and solid solution, a novel complex treatment of plasma nitriding and solid solution was developed, and the research goal is to obtain deep nitrogen-containing layer by this complex treatment and evaluate the performance of the nitrogen-containing stainless steel.
Experimental Procedures
The material used in this research was 304 austenitic stainless steel with the following chemical compositions (wt. %): 0.07C, 1.00Si, 2.00Mn, 18.15 Cr, 8.5 Ni, and Fe balance. The initial specimens were machined into the size of 10 mm × 10 mm × 5 mm. All the surfaces of specimens were treated by using silicon carbide emery papers of different granulometry to achieve a fine finish, and ultrasonically cleaned in dehydrated ethanol for 10 min and dried before plasma nitriding.
Plasma nitriding treatment (referred to as PN) was conducted in a type LD-8CL D.C. plasma nitriding unit with a 24% N2 + 76% H2 atmosphere and gas pressure of 400 Pa at 560 ℃ for 8 h, then the nitrided samples were treated by solid solution (referred to as SS) at 1050 ℃ for 10 h, followed by water quenching.
The phase constituents were determined by X-ray diffraction (XRD) with Cu-Kα (λ¼ 1.54 Å) radiation with scan rate of 0.2°/min, 2θ ranging from 20° to 90°. Hardness measurements were measured by HXD-1000TMC micro-hardness tester with the test load of 25 g and the holding duration of 15 s. Corrosion behavior was evaluated by the potentiodynamic polarization curves in 3.5% NaCl aerated solution using ZAHNER IM6e electrochemical workstations.
Results and Discussions
XRD analysis
Figure 1 shows the XRD patterns of samples untreated and treated by different processes. It clearly illustrates that only typical peaks corresponding to γ phase exist for untreated sample, and the phase compositions are consisted of ε-Fe2,3N, γ'-Fe4N and CrN for plasma nitriding (PN) sample, while only typical peaks corresponding to γN phase exist for PN+SS sample. Therefore, it can be concluded that the nitrides formed during PN process are decomposed by the subsequent solid solution treatment. And the embedded Figure 1 presents that γN has lower diffraction angles than those of γ for the untreated sample.
Micro-hardness analysis and tribological behavior
Figure 2 shows the micro-hardness profile of samples untreated and treated by the complex process. It can be seen that the PN+SS sample owns higher surface hardness and deep effective hardening layer. The surface hardness of the specimen treated by PT+SS is 295 HV0.025, improved 85 HV0.025 compared with that of untreated sample. Meanwhile, the effective hardening layer of the specimen treated by PT+SS reaches 1.1 mm. The increase in hardness could be attributed to the formation of γN phase as shown in Figure 1, which is a supersaturated solid solution of nitrogen in the austenite, hardness improvement by solid solution strengthening by nitrogen atoms.
Corrosion behavior
Figure 3 shows the potentiodynamic polarization curves of samples treated by different processes. It is obvious that the PN+SS sample owns the highest corrosion potential, with higher corrosion potential of -0.473 V and lower corrosion current of 1.045 × 10-6 A/cm2. The polarization curve of the PN+SS sample illustrates that the passive behavior due to the formation of the nitrogen expanded austenite (γN), the incorporation of nitrogen in the interstitial position of the austenite can improved the pitting corrosion resistance of deep nitriding austenitic stainless steel.
Discussion
From the above results, it can be concluded that deep nitrogen-containing layer of stainless steel was formed by the novel combination of plasma nitriding and solid solution. The first step of plasma nitriding is used to get high content of nitrogen in the surface layer of austenitic stainless steel, and solid solution is used to fully decompose the chromium nitrides formed during plasma nitriding and let nitrogen atoms dissolve into the matrix, and thus nitrogen-containing stainless steel can be prepared.
XRD patterns shows that γN has lower diffraction angles than those of γ for the untreated sample due to nitrogen atoms dissolving in the interstitial position of the face centered cubic (fcc) austenite, thus bringing out the shift towards lower angles according to the Bragg Law.
As was known that the addition of nitrogen can improve the hardness of austenitic stainless steel through solution strengthening of interstitial nitrogen, and the corrosion resistance can also be enhanced due to the increase of passive film’s stability by the addition of nitrogen [14], therefore, higher hardness and better corrosion resistance were obtained for the prepared nitrogen-containing stainless steel.
Conclusions
1. Austenitic stainless steel with deep nitrogen-containing layer was prepared by a novel complex treatment of plasma nitriding and solid solution combination.
2. The deep nitrogen-containing layer was composed of nitrogen expanded austenite (γN) layer, and γN has lower diffraction angles than those of γ for the untreated sample.
3. The austenitic stainless steel with deep nitrogen-containing layer has higher hardness and better corrosion resistance, with higher corrosion potential and lower current density in comparison with those of the as-received sample.
Acknowledgments
This research was supported by National Natural Science Foundation of China (51774052) and PAPD of Jiangsu Higher Education Institutions.