
International Journal of Experimental Spectroscopic Techniques
(ISSN: 2631-505X)
Volume 1, Issue 2
Research Article
DOI: 10.35840/2631-505X/8506
Investigating Coffee Samples by Raman Spectroscopy for Quality Control - Preliminary Study
Rafael Carlos Eloy Dias* and Chahan Yeretzian
Table of Content
Figures

Figure 1: Typical Raman spectra (Raman intensity [arb. units] x...
Typical Raman spectra (Raman intensity [arb. units] x relative wavenumber/cm-1) obtained at 1064 nm for samples of roasted coffee beans at different degrees of roasting and laser power.
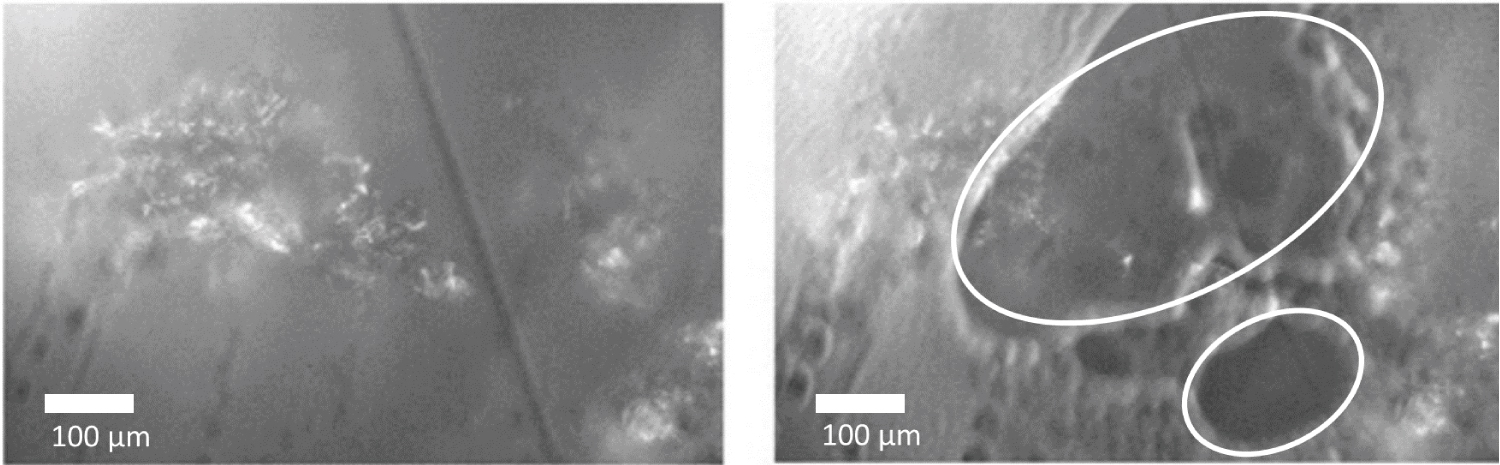
Figure 2: Images of a roasted (light degree) coffeegranule before...
Images of a roasted (light degree) coffeegranule before (on left) and after (on right) the application of 671 nm Raman laser. The circular white marks highlight the damage caused by the laser when the sample is moved in x-y directions. (The dark line on the left image is just a scratch on the support window).

Figure 3: 15Raman spectra (Raman intensity [arb. units] x relative...
Raman spectra (Raman intensity [arb. units] x relative wavenumber/cm-1) at 532 nm for different regions (1-8) of the green coffee bean cross section (right).
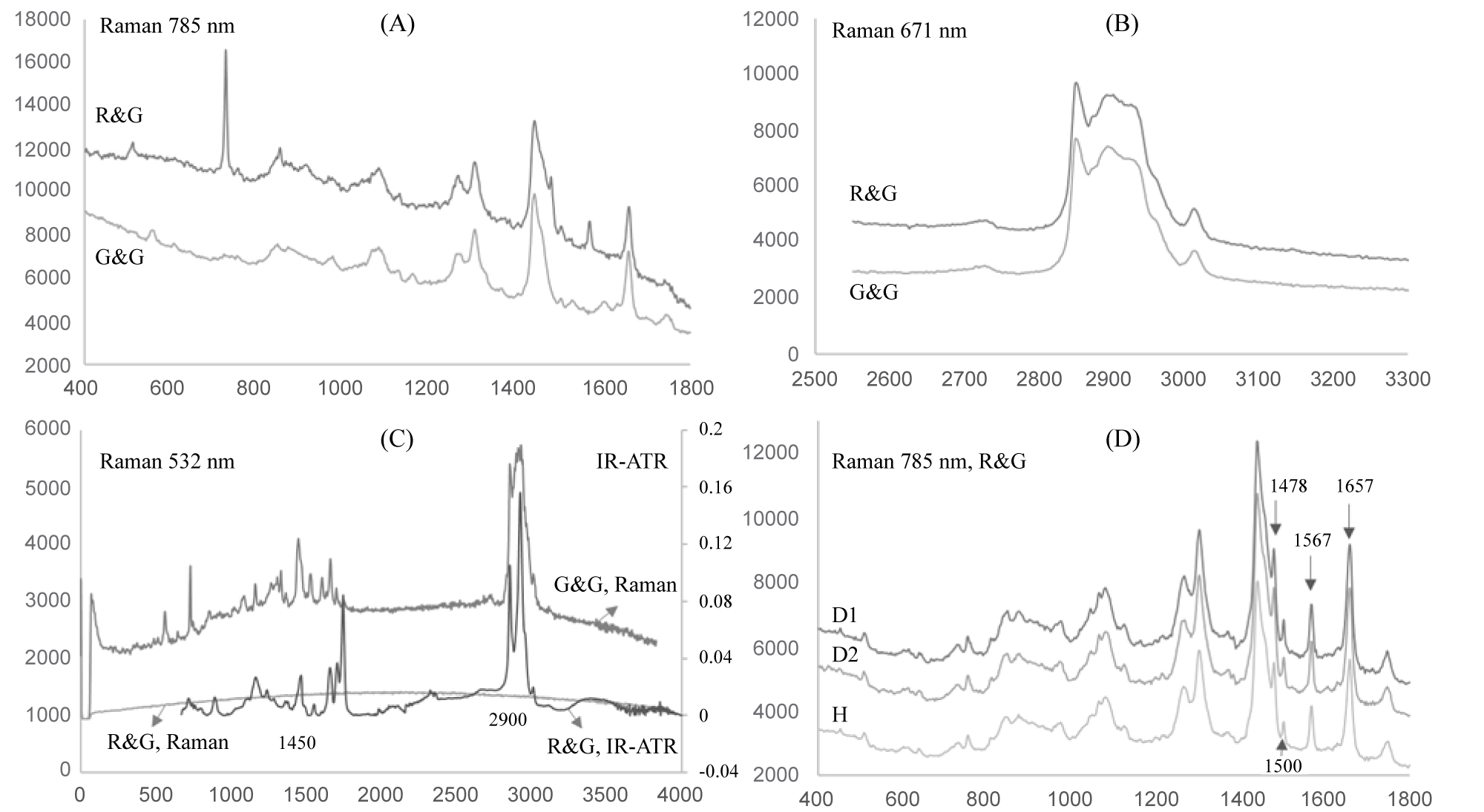
Figure 4: Raman spectra (Raman intensity [arb. units] x...
Raman spectra (Raman intensity [arb. units] x relative wavenumber/cm-1) and FTIR-ATR spectrum (normalized absorbance x wavenumber/cm-1) of coffee oil samples. R&G is the oil of healthy roasted and ground coffee. G&G is the oil of healthy green and ground coffee. D1 and D2 are the oil of roasted and ground samples composed by 70% of healthy beans and 30 % of blends that contain defective beans. H is the oil of roasted and ground healthy beans. All the samples were obtained from Coffea arabica. Medium roasting degree.
References
- Pendergrast M (2000) Uncommon Grounds: The history of coffee and how it transformed our world. Basic Books, New York, 520p.
- Pendergrast M (2009) Coffee second only to oil? Is coffee really the second largest commodity? Tea & Coffee Trade Journal 4.
- Roberto A Buffo, Claudio Cardelli-Freire (2004) Coffee flavour: an overview. Flavour and Fragrance Journal 19: 99-104.
- Grosch W (1998) Flavour of coffee. A review. Nahrung 6: 344-350.
- Farah A (2012) In: C Yi-Fang, Coffee: Emerging Health Effects and Disease Prevention. John Wiley & Sons, Blackwell Publishing Ltd, USA, 21.
- Nkondjock A (2009) Coffee consumption and the risk of cancer: an overview. CancerLett 277: 121-125.
- Petra Huber, Christian Adlhart, Vera Luginbuhl, Fabienne Morf, Sebastian Opitz, et al.(2014) Coffee based polyphenols with potential in skin care: antioxidant activity and skin penetration assessed by in vivo Raman spectroscopy. H&PC - Household and Personal Care 9: 28-34.
- Jean F Grollier, Sophie Plessis (1998) Use of coffee bean oil as a sun filter. US Patent 4793990.
- Marto J, Gouveia LF, Chiari BG, Paiva A, V Isaac, et al. (2016) The green generation of sunscreens: using coffee industrial sub-products. Industrial Crops and Products 80: 93-100.
- Dariusz Dziki, Gawlik-Dziki, Urszula Pecio, Lukasz Rozylo, Renata Swieca, et al. (2015) Ground green coffee beans as a functional food supplement- preliminary study. LWT - Food Science and Technology 63: 691-699.
- Elis Daiane Pauli, Franciele Barbieri, Patricia Salomao Garcia, Tiago Bervelieri Madeira, Vinicius Ricardo Acquaro Junior, et al. (2014) Detection of ground roasted coffee adulteration with roasted soybean and wheat. Food Research International 61: 112 -119.
- Wermelinger T, D'ambrosio L, Klopprogge B, Yeretzian C (2011) Quantification of the robusta fraction in a coffee blend via Raman spectroscopy: proof of principle. J Agric Food Chem 59: 9074-9079.
- Franca AS, Oliveira LS (2015) Spectroscopic methods for chemometric identification of defective and nondefective coffees. In: Preedy VR, Coffee in Health and Disease Prevention. (1st edn), Academic Press-Elsevier, London, UK, 1046.
- Alves RC, Casal S, Alves MR, Oliveira MB (2009) Discrimination between arabica and robusta coffee species on the basis of their tocopherol profiles. Food Chemistry 114: 295-299.
- Gerard Downey, Romain Briandet, Reginald H Wilson, E Katherine Kemsley (1997) Near and mid-infrared (NIR and MIR) spectroscopies in food authentication: the example of coffee varietal identification. J Agric Food Chem 45: 4357-4361.
- Esteban-Diez I ,Gonzalez-Saiz JM, Saenz-Gonzalez C, Pizarro C (2007) Coffee varietal differentiation based on near infrared spectroscopy. Talanta 71: 221-229.
- Moreira I, Scarminio IS (2013) Chemometric discrimination of genetically modified Coffea arabica cultivars using spectroscopic and chromatographic fingerprints. Talanta 107: 416- 422.
- Nádia Reis, Adriana S Franca, Leandro S Oliveira (2013) Discrimination between roastedcoffee, roasted corn and coffee husks by Diffuse Reflectance Infrared Fourier Transform Spectroscopy. LWT- Food Science and Technology 115: 715 -722.
- Ebrahimi-Najafabadi H, Leardi R, Oliveri P, Casolino MC, Jalali-Heravi M, et al. (2012) Detection of addition of barley to coffee using near infrared spectroscopy and chemometric techniques. Talanta 99: 175-179.
- Gordillo-Delgado F, Marin E, Cortes-Hernández DM, Mejia-Morales C, Garcia-Salcedo AJ (2012) Discrimination of organic coffee via Fourier transform infrared-photoacoustic spectroscopy. J Sci Food Agric 92: 2316- 2319.
- Rubayiza AB, Meurens M (2005) Chemical discrimination of arabica and robusta coffees by Fourier Transform Raman Spectroscopy. J Agric Food Chem 53: 4654-4659.
- Keidel A, Stetten D, V Rodrigues C, Máguas C, Hildebrandt P (2010) Discrimination of green Arabica and Robusta coffee beans by Raman spectroscopy. J Agric Food Chem 58: 11187-11192.
- Rasha M El-Abassy, Patrice Donfack, Arnulf Materny (2011) Discrimination between Arabica and Robusta green coffee using visible micro Raman spectroscopy and chemometric analysis. Food Chemistry 126: 1443-1448.
- Richard L Mc Creery (2000) Raman spectroscopyfor chemicalanalysis. JohnWiley & Sons, New York, USA, 420 p.
- Rasha M El-Abassy, Bernd von der Kammer, Arnulf Materny (2015) UV Raman spectroscopy for the characterization of strongly fluorescing beverages. LWT - Food Science and Technology 64: 56-60.
- Joel M Friedman, Robin M Hochstrasser (1975) The use of fluorescence quenchers in resonance Raman spectroscopy. Chemical Physics Letters 33: 225-227.
- Rafael CE Dias, Marta de T Benassi (2015) Discrimination between Arabica and Robusta coffees using hydrosoluble compounds: is the efficiency of the parameters dependent on the roast degree. Beverages 1: 127-139.
- Adriana S Franca, Leandro S Oliveira, Rafael CS Oliveira, Pamela C Mancha Agresti, Rodinei Augusti (2009) A preliminary evaluation of the effect of processing temperature on coffee roasting degree assessment. Journal of Food Engineering 92: 345-352.
- Ana Paula Craig, Adriana S Franca, S Leandro Oliveira (2012) Evaluation of the potential of FTIR and chemometrics for separation between defective and non-defective coffees. Food Chemistry 132: 1368 -1374.
- Ana Paula Craig, Adriana S Franca, Leandro S Oliveira (2012) Discrimination between defective and non-defective roasted coffees by diffuse reflectance infrared Fourier transform spectroscopy. LWT - Food Science and Technology 47: 505-511.
- Campanha, Fernanda Goncalves Dias, Benassi, Rafael Carlos Eloy, Marta de Toledo (2010) Discrimination of coffee species using kahweol and cafestol: effects of roasting and of defects. Coffee Science 5: 87-96.
Author Details
Rafael Carlos Eloy Dias* and Chahan Yeretzian
Institute of Chemistry and Biotechnology, Zurich University of Applied Sciences, SwitzerlandCorresponding author
Rafael Carlos Eloy Dias, Institute of Chemistry and Biotechnology, Zurich University of Applied Sciences ZHAW, Einsiedlerstrasse 31CH - 8820 Wadenswil, Switzerland, E-mail: [email protected], [email protected]
Accepted: April 01, 2016 | Published Online: April 04, 2016
Citation: Dias RCE, Yeretzian C (2016) Investigating Coffee Samples by Raman Spectroscopy for Quality Control - Preliminary Study. Int J Exp Spectroscopic Tech 1:006.
Copyright: © 2016 Dias RCE, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Coffee is a global agricultural commodity of major importance to producing but also to consuming countries world wide. Considering that quality is one of the main factors for price setting, objective processes to assess the quality is in great demand and would be highly appreciated by all players involved in the value chain of coffee. This is particularly relevant for the growing specialty coffee segment. Optical spectroscopic methods have proven to be a valuable approach in the quality control of food products as they are easy to apply, fast and do not require the use chemicals. Here we have explored Raman spectroscopy in order to evaluate and compare different samples of coffee. The aim was to obtain an overview towards developing a tool for quality control. The wavelengths of the Raman laser (532, 671, 785 and 1064 nm) and the potential (50, 100 and 300 mV for analysis at 1064 nm) were varied. Images of the beans were registered during Raman analysis. The main difficulty was to avoid fluorescence interferences in roasted and ground (R&G) coffee. Roasted beans also suffer from the decomposition caused by the Raman laser. Fluorescence was decreased using lower laser power, which, however, also led to a reduction in the intensity of the Raman signals. It was possible to obtain useful Raman responses from green coffee, but which depend on the region of the transversal cross section of the bean. The water and oil extracts of R&G coffee did not generate useful spectra. The best characteristic spectrum was obtained from the oil of ground green coffee, which yielded well-defined peaks and low fluorescence interference, allowing for a better link between the Raman spectrum and the composition of coffee.
Keywords
Roasted and ground coffee, Coffee oil, Green coffee, Quality control, Raman technique
Introduction
Coffee is the second most valuable commodity exported by developing countries, second to crude oil [1,2]. The main product is the roasted and ground coffee, including the rapidly growing segment of specialty or gourmet coffee that is striving for highest quality standards. Consumers are looking for specific characteristics of the coffee beverages, such as special flavors and bodies, which reveals a growing tendency and a shift towards specialty qualities.
In recent years, new coffee-based product formats have been explored. For example, coffee oil, which makes about 10% of the roasted beans weight, is a good matrix for the coffee aroma [3,4]. Hence, coffee oil is being used as a matrix to collect and re-incorporate recovered coffee aroma either in premium soluble coffee or other food products. Furthermore, chlorogenic acids extract, which make up between 4 to 11% of the green coffee bean dry weight [5] and known for their antioxidant properties [6] is used e.g. as a suplement in cosmetic products [7]. Recently new sunscreens have been formulated based on such coffee extracts [8,9]. The use of green coffee as a food supplement is one of the latest developments [10].
Coffee can be produced from different species, geographical origins, with the presence or absence of defects, at several degrees of roasting, and may even contain contaminants [11]. Changes in the quality of the raw material and in the process that coffee is subjected to will result in variations in the composition of the product and its properties. Quality control therefore becomes necessary for the standardization of the final product, which is of interest to consumers, industry and regulatory agencies.
In sensory assessments, even trained tasters may have a wide spread of sensory scores uncertainties; e.g. the assessment of the robusta content in a blend can hardly be achieved with an uncertainty smaller than 20% [12]; therefore instrumental and quatitative evaluation methods are in demand. Several classical techniques are in use for coffee quality control. In spite of the fact that these methods are precise and sensitive, they are often expensive, time and chemical consuming. In many cases, online control or random sampling are impraticable. Therefore, significant effort has been devoted to the development of less onerous and simpler methods. Spectroscopic techniques are here an interesting option [13].
Medium and Near Infrared spectroscopy were used for species discrimination [14-16], and the differentiation of Coffea arabica cultivars [17]. The quality control regarding the defects and impurity of the coffee was also studied using NIR and Diffuse Reflectance [18,19]. Defects is a term used in commercial practice in reference to defective beans, such as sour or brown, black, immature, insect-damaged or worm-holes, as well as extraneous matter, such as woods, husks, skins, and stones [13].
Organic and non-organic R&G coffee were discriminated by infrared-photoacoustic [20]. For the identification of discrepancies among the spectra of each sample, a statistical approach was necessary. Spectroscopic techniques associated to chemometric methods for multivariate analysis, such as PCA (Principal Component Analysis) and PLS (Regression Partial Least Squares), have shown promise in coffee quality monitoring [12,21-23]. In this way, Raman becomes an attractive technique for the evaluation of coffee.
Raman spectroscopy is considered a reliable, simple (to operate) and fast technique. It uses a small quantity of sample and is often nondestrutive (depending on the laser power, as reported here). The spectrum obtained gives information about the structure and composition of solids, liquids, and gases, and is unique for each sample. The Raman effect is based on the inelastic scattering of monochromatic laser radiation by molecular vibration when the scattering is accompanied by a change of polarizability in the chemical bonds. Since the energy losses (frequency shifts) reflect the internal vibrational energies of the molecules in a sample, and the intensity of scattering is directly proportional to the concentration of these molecules, the Raman spectrum, which aggregates the effects of all the different functional groups, is considered a fingerprint of the sample [13,21,24].
Despite the fact that the use of Raman spectroscopy on coffee has been increasing in recent years, especially for the oil of roasted and ground coffee [12,21], for green beans [22,23], the oil of ground green coffee [23], and for water extracts [25], it is still considered an emerging technique for coffee quality control, especially for R&G samples. In particular we are not aware of any study where roasted coffee samples were compared based on Raman spectra. This study explored different samples of coffee to demonstrate the advantages and difficulties of the evaluation of coffee by Raman spectroscopy. We consider that this is an important step for the development of a coffee quality monitoring method based on this technique.
Materials and Methods
Samples of green and roasted coffee, their oil extracts, and water extract of roasted and ground coffee were evaluated. The main variations on the method conditions were the wavelength (532, 671, 785 and 1064 nm) and the power of the Raman laser (50, 100 and 300 mV for analysis at 1064 nm). The spectra were recorded in the range [0 - 4000 cm-1] for the assays at 532 nm and 1064 nm, and [2600 - 4000 cm-1] for tests at 671 nm and 785 nm. In addition to this, a microscope coupled to the Raman equipment was used for imaging purposes, which provided additional information about the sample structure during analysis.
Coffea arabica beans were supplied by Instituto Agronômico do Paraná - IAPAR (Londrina, Brazil: Latitude -23.29, Longitude -51.17; 23° 17' 34" S, 51° 10' 24" W; humid subtropical climate).
For the green sample analysis, a transversal cut was made in the beans with a scalpel. As the coffee bean has different sections, with different compositions [23], several regions of the slice were evaluated, by a motorised computer controlled sample stage. For the ground green beans oil extraction, a green coffee grinder Retsch (Haan, Germany), model MM400, digital, 3 - 30 Hz, was used (1 min, 30 Hz).
To roast the samples to light and medium degrees (corresponding to weight loss of 13% and 17%, respectively), a Probat PRG1Z sample roaster (Emmerich am Rhein, Germany), with a capacity of 100 g, at a maximum of 200°C, was used. Rosting times were 6 to 7 min. Roasted coffee was ground using a Ditting Maschinen AG grinder (Bachenbulach, Switzerland), model KR805, operated at a grinding degree setting of 2. The water extracts were obtained by adding hot tap-water (92°C) to roasted and ground coffee (light degree of roasting). After 3 min, the solution was filtered (paper filter Hario V60; Tokyo, Japan). Two concentrations, 25 and 50 g•L-1, were used. The water extract analyses were performed in small NMR cups (Rotilabo-NMR-Rohrchen) on a glass slide by moving the focus point on the sample.
The oil of two blends of defective beans with healthy beans (roasted at medium level) was used to evaluate the possibility of Raman analysis in classifying coffee samples. The samples (D1 and D2) were composed of 70 % (w/w) of healthy Coffea arabica beans and 30 % of a blend (Blend 1 and Blend 2) of defective beans with healthy beans (C. arabica). These blends were classified by a trained and experienced professional (from IAPAR) as following: Blend 1 (that composed 30 % ofD1) - whole-healthy (10.6 %), broken (37.4 %), sour (37.5 %), black (13.2 %), and skin+woods (1.4 %); Blend 2 (that composed 30 % of D2) - whole-healthy (11.1 %), broken (28.2 %), sour (53.5 %), black (6.0 %), and skin+woods (0.7 %). These samples (Blend 1 and Blend 2) were obtained from coffee farmers in the region of IAPAR, Brazil, i.e., they were not assembled in a laboratory.
To obtain the coffee oils, the extraction was carried out (10 g of ground sample with 20 g of sodium sulfate, a drying agent) by Soxhlet (Buchi, model B-811; Flawil, Switzerland) for 3 h with tert-butyl methyl ether (modified from [12]).
For Raman analysis, three different pieces of equipment were tested: i) Confocal Raman microscope Alpha 300, WITec (Ulm, Germany) equipped with a green laser at 532 nm, 20x/50x/100x objectives, and a microscope (WITec) x-y-z scan stage; ii) Skin Analyzer - River Diagnostics BV (Rotterdam, Netherland), model 3510 SCA, coupled to an inverted microscope DERM 1000 measurement x-z stage (785 nm and 671 nm lasers); iii) FT-Raman spectrophotometer (Bruker, Billerica, USA), model RFS 100/S, equipped with a laser of 1064 nm (laser control AD LAS, Nd:YAG laser), coupled with a manual biological microscope Nikon Optiphot (Tokyo, Japan). No calculation was made. The spectra displayed here were obtained from the raw data. A Fourier transform infrared spectrometer with attenuated total reflectance detector (FTIR-ATR) (Bruker, Billerica, EUA, Tensor 37, with a cell of analysis Specac, Orpington, England) was used to compare coffee oil samples with the Raman analysis. The spectrum was obtained by 10 seconds-reading. An oil drop was analyzed (in triplicate) directly on the window of reading.
Discussion/Conclusion
Raman signals were not detected for the water extracts. For roasted and ground coffee at 1064 nm, an increase in the Raman response with decreasing wavenumber (Figure 1) was observed, which is typical of fluorescence interference for dark samples [25].
The difficulty in identifying peaks that could be related to the sample composition at higher laser power (300 mV) is notable (Figure 1). In fact, Raman intensity is proportional to the laser power, but so is the fluorescence [24]. There are some propositions in the literature as to how to avoid these interferences, such as the use of QuEChERS [26] or by changing the laser wavelengths [25]. However, no report was found describing the direct analysis of R&G coffee by Raman technique.
The roasting degree has little effect on the Raman responses, as exemplified in figure 1 (light to medium roast at the same laser power). However, damage caused by the heating from the laser (671 nm) was verified on the coffee bean surface (Figure 2). The roasted bean absorbed the laser light energy that is supplied through the lens to a very small spot on the sample. This means that the sample heats up locally and eventually get darker and absorb even more light. The result is damage on the sample. This is also a limitation of Raman analysis for roasted coffee, since it is important to preserve the structure of the sample to obtain reliable results.
For green coffee at 532 nm, the spectral profile varied with the bean's cross-sectional region, especially in terms of fluorescence interference, which is illustrated by the parabolic tendency (Figura 3). Regions 1, 4 and 5 of the coffee bean are visually similar, but differ in the spectral profile. Similar spectra were expected for areas 2 and 8, which are border regions of the bean, but which could not be verified in the data. Moreover, regions 3 and 7, which are from different regions (between center and border, and in the center, respectively), produced similar profiles, considering the peaks and the fluorescence interference (Figura 3). The fact that we observed variations of the spectra by varying the region of the green bean makes it difficult to standardize the Raman analysis for these samples. In 2011, El-Abassy et al. [23] reported on Raman analysis of three different layers of the green Coffea arabica surface slices. They highlighted discrepancies in the intensities of Raman peaks (514.5 nm) particularly around 1600 cm-1, which were attributed to the heterogeneity of distribution of chlorogenic acids and lipids in the layers. While in this work and setup variations were observed on different regions of an x-y-z surface of a green bean layer, El-Abassy et al. examined different layers. Both studies are consistent and confirm that Raman spectra vary for different spots and regions within the coffee green structure. This is likely due to the 3D inhomogeneity in the chemical structure and composition of green beans.
Figure 4 presents the oil spectra of roasted and ground coffee (R&G) and green and ground coffee (G&G) at different wavelengths. It was compared the Raman spectra for both sample (healthy beans) at 785 nm, between 400-1800 cm-1 (Figure 4A). At 671 nm, the same samples were evaluated, but in the region 2550-3300 cm-1 (Figure 4B). The range between 2800 and 3100 cm-1 are attributed to symmetric and asymmetric C-C and C-H stretching vibrations, while the signals between 1200 and 1800 cm-1 are related to typical organic groups, which are more relevant to the discrimination of coffee species and considered the fingerprint of the samples [12,21,22]. Considering that roasting is an intense process that induces physical transformations, a large range of chemical reactions and important changes in the coffee composition [27,28], it is to be expected that changes occur in the lipid fraction as well. However, it can be observed that G&G and R&G oils have a similar Raman spectra profile, with subtle differences (Figure 4A and Figure 4B).
At 532 nm, no Raman signal was obtained with respect to R&G coffee oil (Figure 4C), probably due to the interference of pigments and other compounds developed during the roasting process, which was not observed at other wavelengths. The Raman spectra for G&G oil at 532 nm showed an interesting profile, matching the literature results [21]. Of additional interest is the comparison between this spectrum and the FTIR-ATR (Fourier transform spectroscopy with attenuated total reflectance detector) spectrum of R&G coffee (Figure 4C). It is possible to verify similarities in the wavenumber of the signals and in the shape of the peaks, which demonstrate similarities in the information about chemical groups, such as the peaks around 1450 cm-1 and 2900 cm-1. It indicates that Raman analysis at 532 nm can provide reliable information about the sample composition before the roasting process, once the infrared Fourier transform spectroscopy with reflectance detector has become a well-established technique for roasted coffee samples discrimination [29,30].
One possible application of Raman analysis for coffee quality control is the determination of the defects ratio in a blend. After the roasting and grinding processes, chemical-based assessment is necessary to detect the defective beans, since the visual and mechanical methods are no longer applicable. Two samples composed of blends of defects with healthy beans (D1 and D2) were evaluated at 785 nm in the Raman fingerprint region. Roasted and ground under the same conditions, the samples presented characteristic Raman spectrum profiles. The Raman scattering bands attributed to cafestol and kaweol [21], two diterpenes present in the unsaponifiable matter of coffee lipids, were observed. Bands at 1478, 1500 and 1567 cm-1, related to C = C stretching of furan cycle, and at 1657 cm-1, related to C = C streching of cyclohexene, were detected (Figure 4D). Diterpenes are considered coffee species discriminators since they are present in different concentrations when Coffea arabica and C. Canephora are compared [31]. D1 and D2 have different proportions in terms of defects. Further evaluations using chemometric techniques for the data analysis have to be done to verify if the slight differences visually observed can explain the differences in terms of defects. The spectra in figure 4D were normalized to compensate for any change in experimental conditions.
Slight differences can be observed in the shape and absorbance of the bands in figure 4D. It means that these results are assessable by chemometric methods for the investigation of similarities and discrepancies among the samples, which could classify the different samples of coffee in terms of defects.
Considering the above discussion, we conclude that further study should be conducted regarding to the direct Raman analysis of R&G coffee. Extra care should be taken when the Raman spectrum is acquired from green coffee, since the response is dependent on the bean surface area being analysed. The Raman spectra of R&G and G&G oils provide reliable information about the sample composition. Raman spectroscopy in conjunction with an appropriate statistical method can be used for the development of a tool for coffee quality monitoring.
Acknowledgements
We acknowledge Science without Borders, a Brazilian government program for students abroad, for the financial support, IAPAR, for providing the sample, Michael J. Reber, for the review of English, and Christian Adlhart and Michael Edelmann for the support in the analysis.

