Development and Validation of an Enzyme-Linked Immunosorbent Assay for the Measurement of Human Coagulation Factor XI in Human Immunoglobulin Preparations

International Journal of Analytical and Bioanalytical Methods
(ISSN: 2633-8912)
Volume 3, Issue 1
Original Research
DOI: 10.35840/2633-8912/2415
Development and Validation of an Enzyme-Linked Immunosorbent Assay for the Measurement of Human Coagulation Factor XI in Human Immunoglobulin Preparations
Andrea Engelmaier and Alfred Weber
Table of Content
Figures
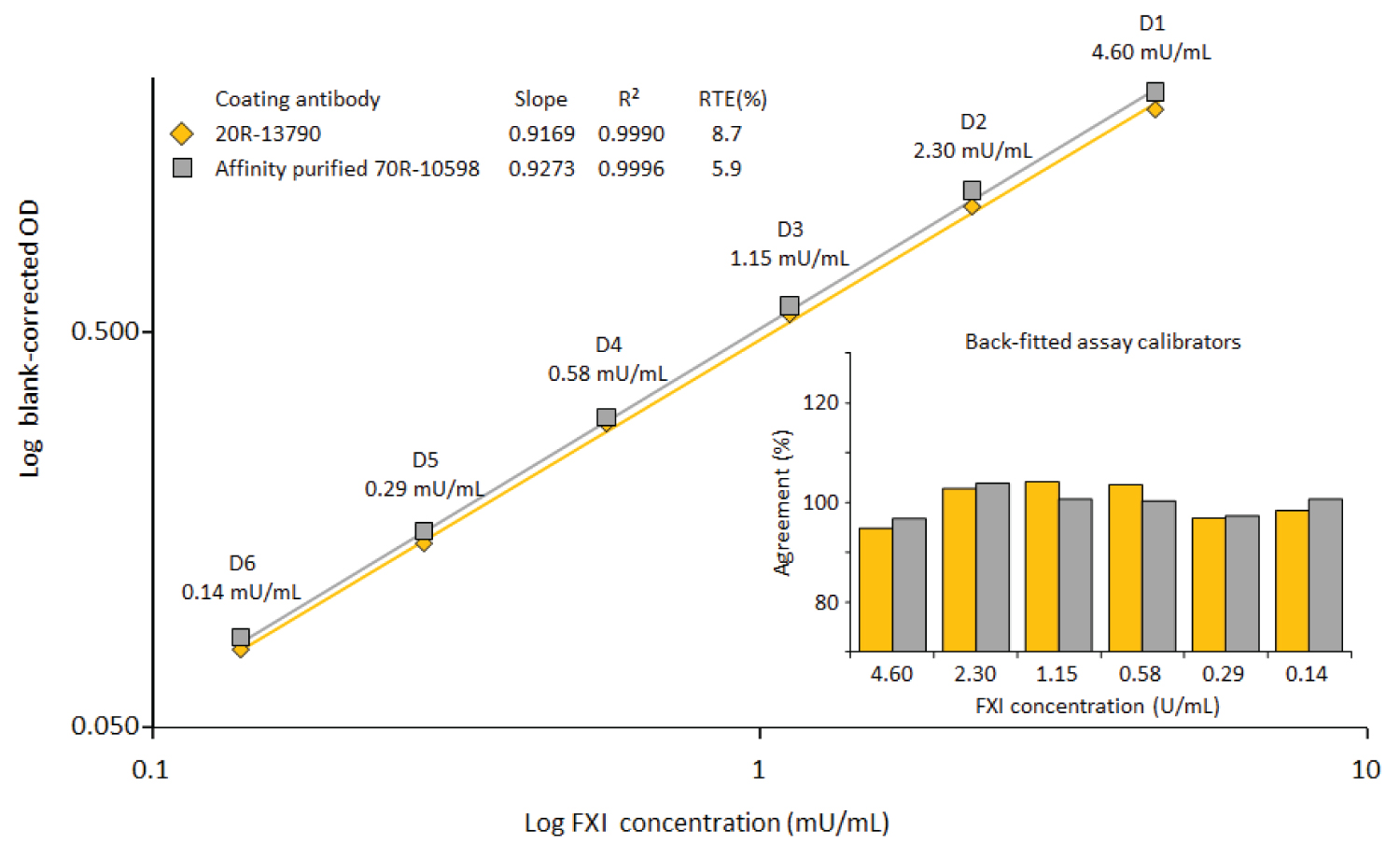
Figure 1: Comparison of two commercially available....
Comparison of two commercially available anti-FXI IgG preparations.
Both anti-FXI preparations were coated at 10 μg/mL. A reference plasma preparation was used for the construction of the log-log calibration curves, ranging from 0.14 to 4.6 mU FXI/mL. The insert shows the agreement of the back-fitted assay calibrators with their nominal concentrations.

Figure 2: Differentiation between FXI and.....
Differentiation between FXI and FXIa.
Panel A shows the dilution-response curves obtained for the reference plasma preparation used as the assay standard and FXI spiked to buffer, FXI-deficient and FXI-depleted plasma. Panel B shows the same samples, spiked with FXIa.
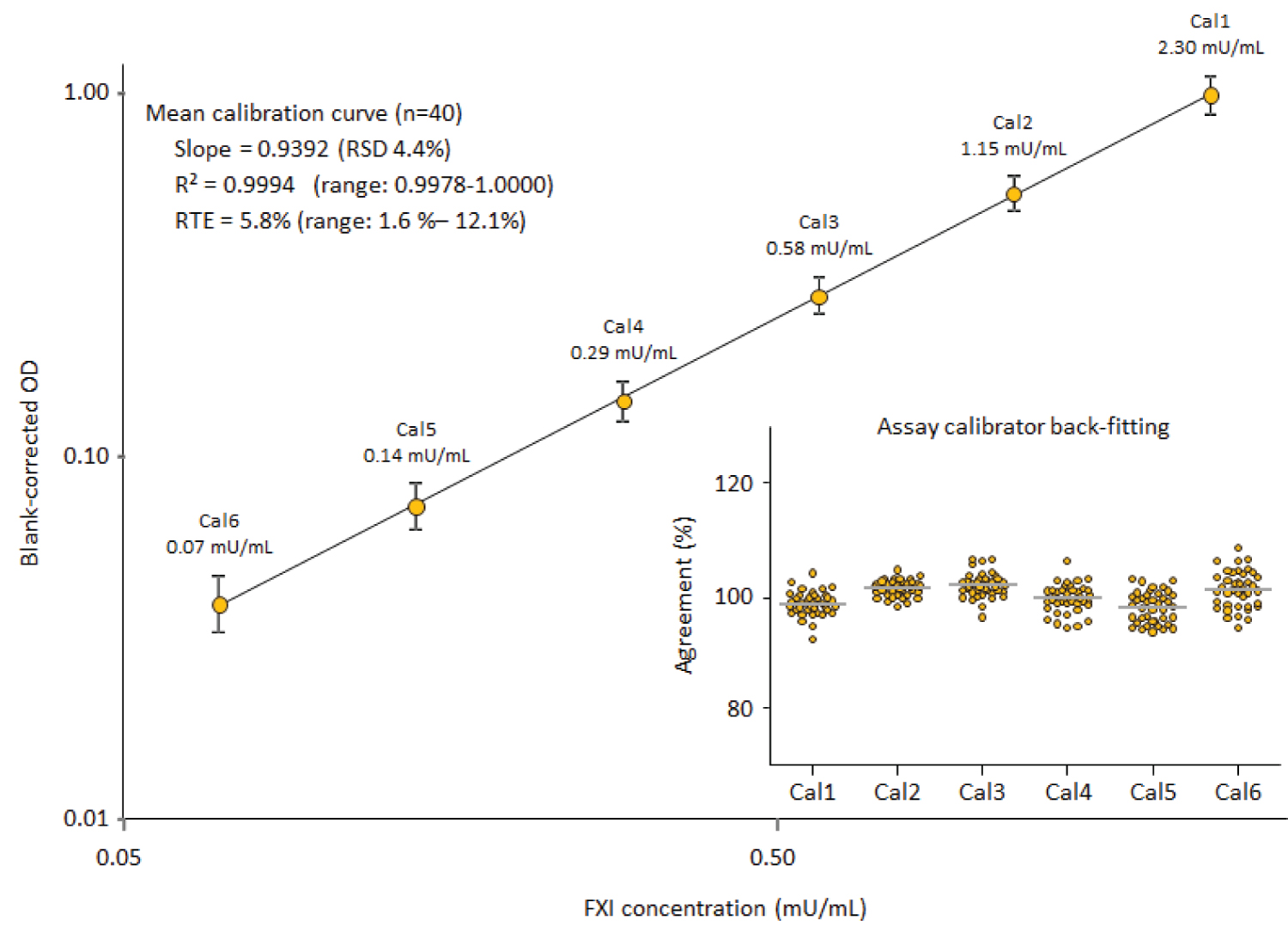
Figure 3: Mean assay calibration curve....
Mean assay calibration curve.
The mean (n = 40) log-log assay calibration curve, ranging from 0.07 to 2.3 mU FXI/mL, is shown together with its mean characteristics slope, correlation coefficient and relative total error (RTE). The insert shows the agreement of the back-fitted assay calibrators with their nominal concentrations as a measure for the goodness of the calibration curve fitting.
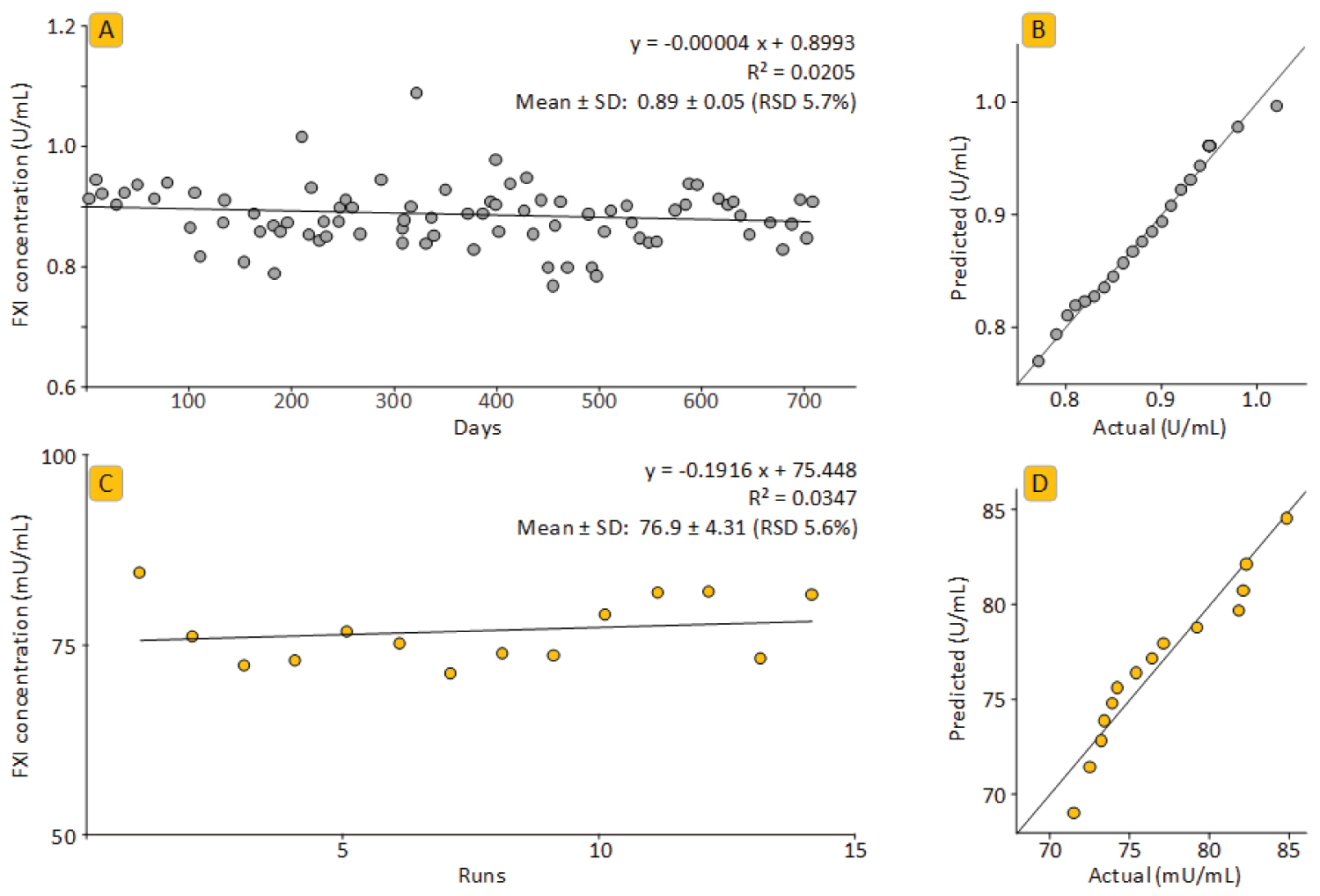
Figure 4: Precision analysis over extended time....
Precision analysis over extended time.
Panels A and B show the results for the plasma control preparation, panels C and D present the data for the repeated analysis of a GAMMAGARD LIQUID preparation. In particular, panels A and C show the FXI concentrations over time, panels B and D show the quantile-quantile plots calculated with GraphPad Prism 8.3.

Figure 5: Linearity investigation for three....
Linearity investigation for three IgG products with different IgG concentrations.
The dilution-response curves for GG-SD, GGL and CUV, measured in their minimum dilutions of 1/40, 1/100 and 1/100, respectively, are shown for the neat and FXI-spiked samples. The corresponding plasma standard curve is also shown.
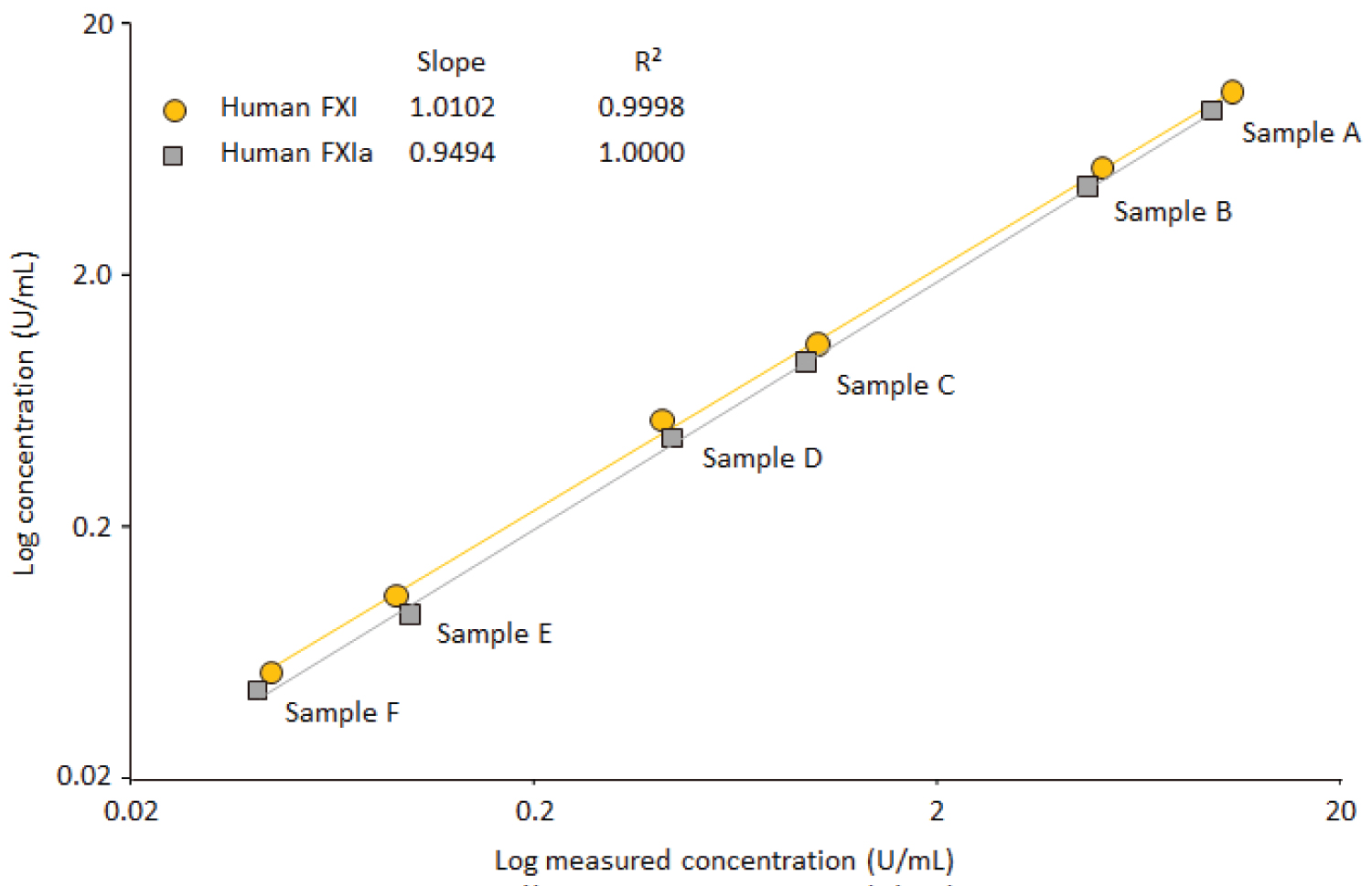
Figure 6: Linearity check for purified FXI and....
Linearity check for purified FXI and FXIa.
Six samples with defined FXI/FXIa concentrations ranging from 0.05 to 10 U FXI/FXI were measured. The regression curves between the concentrations expected and found were calculated. The insert shows the corresponding slopes and correlation coefficients of these curves.
Tables
Table 1: Results for the various coating conditions.
Table 2: Influence of benzamidine.
Table 3: Spike-recovery data for the IgG products GAMMAGARD S-D, GAMMAGARD LIQUID and CUVITRU.
Table 4: FXI results for the international reference preparations.
Table 5: Precision analysis for IgG products and intermediates.
Table 6: Results of the parallelism investigation for the three IgG products.
Table 7: Measurement of the FXI-deficient human plasma.
References
- Bouma BN, Griffin JH (1977) Human blood coagulation factor XI purification, properties, and mechanism of activation by activated factor XII. J Biol Chem 252: 6432-6437.
- Mohammed BM, Matafonov A, Ivanov I, Sun MF, Cheng Q, et al. (2018) An update on factor XI structure and function. Thromb Res 16: 94-105.
- Furie B, Furie BC (1988) The molecular basis of blood coagulation. Cell 53: 505-518.
- Fujikawa K, Chung DW, Hendrickson LE, Davie EW (1986) Amino acid sequence of human factor XI, a blood coagulation factor with four tandem repeats that are highly homologous with plasma prekallikrein. Biochemistry 25: 2417-2424.
- Faid V, Denguir N, Chapuis V, Bihoreau N, Chevreux G (2014) Site-specific N-glycosylation analysis of human factor XI: Identification of a noncanonical NXC glycosite. Proteomics 14: 2460-2470.
- Geng Y, Verhamme I, Smith SB, Sun MF, Matafonov A, et al. (2012) The dimeric structure of factor XI and zymogen activation. Blood 121: 3962-3969.
- Schmaier AH (2016) The contact activation and kallikrein/kinin systems: Pathophysiologic and physiologic activities. J Thromb Haemost 14: 28-39.
- Puy C, Tucker EI, Matafonov A, Cheng Q, Zientek KD, et al. (2015) Activated factor XI increases the procoagulant activity of the extrinsic pathway by inactivating tissue factor pathway inhibitor. Blood 125: 1488-1496.
- Alving BM, Tankersley DL, Mason BL, Rossi F, Aronson DL, et al. (1980) Contact-activated factors: Contaminants of immunoglobulin preparations with coagulant and vasoactive properties. J Lab Clin Med 96: 334-346.
- Wolberg AS, Kon RH, Monroe DM, Hoffman M (2000) Coagulation factor XI is a contaminant in intravenous Immunoglobulin preparations. American J Hematol 65: 30-34.
- (2010) European medicines agency recommends suspension of Octagam in all EU member states.
- Sridhar G, Ekezue BF, Izurieta HS, Selvam N, Ovanesov MV, et al. (2014) Immune globulins and same-day thrombotic events as recorded in a large health care database during 2008 to 2012. Transfusion 54: 2553-2565.
- Etscheid M, Breitner-Ruddock S, Gross S, Hunfeld A, Seitz R, et al. (2012) Identification of kallikrein and FXIa as impurities in therapeutic immunoglobulins: implications for the safety and control of intravenous blood products. Vox Sang 102: 40-46.
- Roemisch J, Zapfl C, Zoechling A, Pock K (2012) Comparison of thrombin generation assay and non-activated partial thromboplastin time for the assessment of enhanced procoagulant activity in immunoglobulin solutions. WebMec Central.
- Seifner A, Beck G, Bayer P, Eichmeir S, Lackner F, et al. (2014) Assessment of immunoglobulin concentrates on thrombogenic activity by thrombin generation assay, prekallikrein activator assay, and size-exclusion chromatography. Transfusion 54: 376-383.
- (2012) European pharmacopeia: Human normal immunoglobulin for intravenous administration. Monograph 2012: 0918.
- Jose M, Marzo N, Pons B, Herrerias A, Lopez L, et al. (2013) Flebogamma DIF (intravenous immunoglobulin) purification process effectively eliminates procoagulant activities. Biologicals 41: 393-399.
- Komenda M, Stadler D, Malinas T, Moses M, Herzog PE, et al. (2014) Assessment of the ability of the Privigen purification process to deplete thrombogenic factor XIa from plasma. Vox Sang 107: 26-36.
- Briseid K, Hoem NO, Johannesen S, Haug K (1995) Amidolytic assay of factor XI in human plasma - significance of kallikrein for the activity measured. Thromb Res 78: 239-250.
- (2021) Biophen FXIa: Chromogenic assay for measuring Factor XIa activity. HYPHEN BioMed.
- (2021) ROX FXIa chromogenic assay information.
- Wilmot HV, Gray E (2020) Enabling accurate measurement of activated factor XI (FXIa) in therapeutic immunoglobulin products. Vox Sang.
- Gray EWH, Hogwood J, Rigsby P (2012) Evaluation of the proposed WHO 1st reference reagent for activated blood coagulation factor XI (FXIa), Human. WHO/BS/2012.2206.
- Gray E, Hogwood J, Wilmot H, Thelwell C, Dougall T, et al. (2014) Value assignment of the candidate 1st international standard for activated blood coagulation factor XI (FXIa), human, NIBSC code 13/100. WHO/BS/2014.2245.
- Liang Y, Jackson JW, Woodle SA, Surov SS, Parunov LA, et al. (2020) Detecting factor XIa in immune globulin products: Commutability of international reference materials for traditional and global hemostasis assays. Res Pract Thromb Haemost.
- (2020) Datasheet for ISTH/SSC#4 reference plasma.
- (2011) European medicines agency: Guideline on bioanalytical assay validation. EMEA/CHMP/EWP/192217/2009.
- (2017) Assignment of potency to the WHO 1st international standard for blood coagulation factor XI in plasma, human (04/102) and calibration of blood coagulation factor XI in SSC secondary plasma standard lot #3. WHO/BS/05.2017.
- Plikaytis BD, Holder PF, Pais LB, Maslanka SE, Gheesling LL, et al. (1994) Determination of parallelism and nonparallelism in bioassay dilution curves. J Clin Microbiol 32: 2441-2447.
- Stiehm ER, Orange JS, Ballow M, Lehman H (2010) Therapeutic use of immunoglobulins. Adv Pediatr 57: 185-218.
- Berger M (2008) Principles of and advances in immunoglobulin replacement therapy for primary immunodeficiency. Immunol Allergy Clin North Am 28: 413-437.
- Chapel HM, Spickett GP, Ericson D, Engl W, Eibl MM, et al. (2000) The comparison of the efficacy and safety of intravenous versus subcutaneous immunoglobulin replacement therapy. J Clin Immunol 20: 94-100.
- Wasserman RL, Melamed I, Stein MR, Gupta S, Puck J, et al. (2012) Recombinant human hyaluronidase-facilitated subcutaneous infusion of human immunoglobulins for primary immunodeficiency. J Allergy Clin Immunol 130: 951-957.
- European Blood Alliance and International Plasma and Fractionation Association (EBAIPFA) Amsterdam, the Netherlands 14-15 January 2020. The Marketing Research Bureau, Inc. Patrick Robert.
- Wilmot H, Hockley J, Rigsby P, Gray E (2017) Establishment of the world health organization 2nd international standard for factor XI, plasma, human. Front Med 4: 28.
- David T, Kim YC, Ely LK, Rondon I, Gao H, et al. (2016) Factor XIa-specific IgG and a reversal agent to probe factor XI function in thrombosis and hemostasis. Sci Transl Med 8: 353ra112.
Author Details
Andrea Engelmaier1 and Alfred Weber2*
1Baxalta Innovations GmbH, a Takeda Company, Pharmaceutical Sciences, Austria
2Baxalta Innovations GmbH, a Takeda Company, Plasma Derived Therapies, Austria
Corresponding author
Alfred Weber, Baxalta Innovations GmbH, a Takeda company, Plasma Derived Therapies, A-1220 Vienna, Industriestraße 67, Vienna, Austria, Tel: +43-1-20100-2472662; +43-664-8120319.
Accepted: May 01, 2021 | Published Online: May 03, 2021
Citation: Engelmaier A, Weber A (2021) Development and Validation of an Enzyme-Linked Immunosorbent Assay for the Measurement of Human Coagulation Factor XI in Human Immunoglobulin Preparations. Int J Analyt Bioanalyt Methods 3:015.
Copyright: © 2021 Engelmaier A, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Following an accumulation of incidences of thromboembolic events after administration of certain immunoglobulin G (IgG) brands in 2010 and 2011, regulatory agencies also required other therapeutically used IgG preparations to be checked for the presence of procoagulant activities. Additionally, their manufacturing pathways had to be investigated for steps that have the capacity to reduce procoagulant activities. Apart from measuring activated factor XI (FXIa) with different methods including thrombin generation, coagulation or chromogenic assays, measuring factor XI (FXI) with an enzyme-linked immunosorbent assay (ELISA) could substantially augment information on the procoagulant potential of an IgG preparation as it would provide information on the total FXI level present, independent of its activation state. The particular sample matrix, consisting of pooled polyclonal human IgG up to 200 mg/mL and showing pH values below 5.0, poses a unique challenge for ELISA development. The data presented describe the development of a FXI ELISA based on using a commercially available polyclonal rabbit anti-human FXI IgG antibody: This preparation was also used for preparing the biotinylated detection antibody. This ELISA does not discriminate between FXIa and FXI protein (zymogen) as a similar response is shown for both forms. Furthermore, the assay validation data obtained are summarized. In particular, the assay attributes accuracy, precision, linearity and specificity were addressed for the three therapeutically used immunoglobulin G preparations GAMMAGARD S/D, GAMMAGARD LIQUID and CUVITRU, showing different IgG levels from 50 to 200 mg/mL. The validation data obtained demonstrated adequate performance characteristics qualifying the FXI ELISA as a sensitive, accurate and handy analytical tool for investigating the procoagulant potential of immunoglobulin G preparations.
Keywords
Factor XI measurement, Therapeutically used immunoglobulin preparations, Procoagulant proteins, Plasma products, ELISA development
Abbreviations
BSA: Bovine Serum Albumin; DB: Dilution Buffer; CUV: 20% subcutaneous immunoglobulin G CUVITRU; ELISA: Enzyme-Linked Immunosorbent Assay; EP: European Pharmacopeia; FXI(a): (activated) Coagulation factor XI; FXIIa: Activated coagulation factor XII; GGL: 10% liquid human intravenous immunoglobulin G preparation GAMMAGARD LIQUID; GG-SD: 5% lyophilized human immunoglobulin G preparation GAMMAGARD S/D; IgG: Immunoglobulin G; IVIG: Intravenous immunoglobulin G; OD: Optical Density; PBS: Phosphate-Buffered Saline; PBST: Phosphate-Buffered Saline with Tween 20; PptG: Intermediate of the GGL manufacturing process; RSD: Relative Standard Deviation; SCIG: Subcutaneous Immunoglobulin G; RTE: Relative Total Error; RT: Room Temperature; SD: Standard Deviation
Introduction
Factor XI (FXI) is a 160-kDa plasma glycoprotein [1,2], present in normal human plasma at a concentration of about 5 μg/mL (= 30 nM) [3]. Structurally, FXI circulates as a homodimer consisting of two identical, disulfide bond-linked subunits with a size of 607 amino acids. Four so-called "apple domains" (A1, A2, A3, A4) each of them comprising 90 or 91 amino acids form the N-terminal part of the molecule, while the typical trypsin-like serine protease domain is located in the C-terminal part. The disulfide bridge, generating the FXI homodimer, is established between the C321 residues, located in the A4 domains of two subunits. Of note, human plasma prekallikrein and FXI share 58% of their amino acid sequence, and both proteins circulate in plasma non-covalently complexed with high-molecular-weight kininogen. In the past, five potential N-glycosylation sites have been identified on the positions N72, N108, N335, N432 and N473 in each of the polypeptide subunits of FXI [4] and recently, site-specific N-glycan analysis has been carried out [5]. After activation, activated FXI (FXIa) [6] is involved in the contact activation of blood coagulation [7]. FXI activation occurs by limited proteolysis of the R369-I370 bond by activated factor XII (FXIIa). Alternatively, FXI activation by thrombin or autoactivation can occur. In the latter cases, FXI needs not be a dimer which is essential for the activation by FXIIa [6]. The serine protease FXIa formed not only selectively activates coagulation factor IX (FXI) as its physiological substrate within the intrinsic coagulation cascade but also increases the procoagulant activity of the extrinsic coagulation pathway by inactivating tissue factor pathway inhibitor [8].
Intravenous and subcutaneous immunoglobulin preparations (IVIG, SCIG), purified from pooled human plasma, are important biotherapeutics. They are essential not only for the lifelong replacement therapy established for primary antibody-deficient patients more than 30 years ago, but also have proven immunomodulatory effects in some autoimmune and inflammatory diseases. The first evidence for the presence of procoagulant and vasoactive substances in IVIG preparations was provided by Alving, et al. [9]. Later, Wolberg and colleagues identified FXI and more specifically FXIa as a procoagulant contaminant in such preparations [10]. The clustered occurrence of thromboembolic adverse events after administration of IVIG was associated with specific brands. Thus, in 2010 IVIG Octagam® (Octapharma) was temporarily removed from the market as its administration could be associated with a rise in thromboembolic events [11]. Increased incidences of thromoboembolic events were further observed for the IVIG brand Omr-IgG-am® (Omrix Biopharmaceuticals) and the SCIG preparation Vivaglobin® (CSL Behring) [12]. Combined scientific efforts of health agencies and manufactures resulted in FXIa being identified as the main causative agent [13-15]. As a consequence, the European Pharmacopeia monograph for IVIG was updated [16] and now states with regard to procoagulant activities: "The method of preparation also includes a step or steps that have been shown to remove thrombosis-generating agents. Emphasis is given to the identification of activated coagulation factors and their zymogens and process steps that may cause their activation". Different manufacturing steps with a well proven capacity to remove thrombosis-generating agents have been reported for specific products [17,18].
Analytically, FXIa can be determined by nonactivated partial thromboplastin time, thrombin generation assays, non-specific FXIa chromogenic assays [19] and, in addition, two specific FXIa chromogenic assays [20-22]. The latter assays attempt to mimic the coagulative FXIa in vivo activity as the generation of activated factor IX is determined in a purified system containing the components of the factor X activating tenase complex, namely activated factor VIII, factor X and factor IX. The finally generated FX activity directly correlates to the FXIa activity present in the test sample. The burst of research on assays for measuring FXIa culminated with two reference materials developed by the United Kingdom's National Institutes for Biological Standards and Controls: NIBSC 11/236 and NIBSC 13/100. These proposed standards were adopted by the World Health Organization (WHO) as the WHO First International Reference Reagent [23] and the WHO First International Standard for FXIa [24], respectively. Their calibration was, however, exclusively based on using chromogenic FXIa assays. Liang, et al. [25] recently provided an alignment for other FXIa assays including nonactivated partial thromboplastin time, microplate clotting and thrombin generation assays for testing IgG products in both normal plasma and FXI-deficient plasma.
Apart from the measuring FXIa activity with different methods, measurement of the FXI zymogen is fostered by the European Pharmacopeia monograph as this approach provides information on the maximum FXIa levels which can be reached given that complete FXI activation can take place. Therefore, we developed an enzyme-linked immunosorbent assay (ELISA), based on using a commercially available polyclonal anti-FXI IgG preparation purposefully designed for the accurate and precise measurement of FXI in the sample matrix of IVIG and SCIG preparations. These matrices are characterized by high concentrations of polyclonal human IgGs and slightly acid pH values below pH 5.0. Both specific characteristics pose challenges for the sensitive ELISA measurement of FXI as high dilutions cannot be applied to overcome these limitations. Furthermore, assay validation data are presented for the measurement of FXI in three different therapeutically used IgG products with different IgG concentrations.
Materials and Methods
Materials
The AssayMax human factor XI ELISA kit (EF1011-1) from Assaypro (St. Charles, Missouri, USA) was tested. The polyclonal rabbit anti-human FXI IgG preparations 20 R1379 and 70 R 10598 (affinity-purified) were from Fitzgerald (Acton, Massachusetts, USA). Streptavidin peroxidase (P0397) was purchased from DakoCytomation (Vienna, Austria) and the fresh frozen reference plasma preparation CRYOcheck from Precision BioLogic (CoaChrom; Vienna, Austria). This preparation, used for the assay calibration, had a labelled FXI concentration of 0.92 U/mL. In addition, the lyophilized reference plasma preparation from Technoclone (Vienna, Austria) was used as an assay control. A patient derived human FXI-deficient plasma was obtained from George King Bio Medical Inc. (Overland Park, Kansas, USA), while the artificially FXI-depleted plasma preparation was from American Diagnostica (Pfungstadt, Germany). The purified human FXI preparation HCXI 0150 and the human FXIa preparation HFXIa were from Haematologic Technologies Inc. (Essex Junction, Vermont, USA) and Enzyme Research Laboratories (South Bend, Indiana, USA), respectively. The ISTH/SSC#4 reference plasma [26] and the FXIa WHO standard 11/236 [23] with labelled FXI and FXIa concentrations of 0.89 U/mL and 10 IU/mL, respectively, were also used. Sulfo-NHS-LC-Biotin reagent 21327 was purchased from Thermo Scientific (Waltham, Massachusetts, USA). The degree of biotinylation was determined with the fluorometric biotin quantification kit 46610 from Thermo Scientific. Maxisorp F96 flat-bottom plates, KCl, NaCl, KH2PO4, Na2HPO4 × 2 H2O, H2SO4 (95-97%), Na2CO3, NaHCO3 and water (LiChrosolv) were obtained from VWR (Vienna, Austria). Tween 20 (EIA grade) was from Bio-Rad (Vienna, Austria), the peroxidase substrate SureBlue from KPL (Medac, Hamburg, Germany) and bovine serum albumin (BSA; A0281) and benzamidine hydrochloride from Sigma (Vienna, Austria). The following IgG preparations, all from Baxter (Vienna, Austria) were used: GAMMAGARD LIQUID/KIOVIG (GGL), GAMMAGARD S/D (GG-SD) and CUVITRU (CUV). Four representative intermediates of the GGL manufacturing process were included in the validation, all derived from one lot: The starting material plasma pool, II+III extract, II+III filtrate and precipitate G (PptG). Aliquots of these samples were prepared and kept frozen at -20 ℃ until measurement. Regularly calibrated equipment was used: Automatic plate washers ELx405 and the ELISA reader EL808 (both from Biotek, Szabo, Vienna, Austria).
Biotinylation of the rabbit anti-FXI antibody
The Sulfo-NHS-LC-Biotin reagent from Thermo Scientific was used following the instructions of the manufacturer. Briefly, 1 mL polyclonal rabbit anti-human FXI 20-R-1379 was diluted with phosphate-buffered saline (PBS) to 2 mg/mL and dialyzed against PBS at 4 ℃ overnight. Then, 70 μL of the 10 mM biotinylation reagent was added to obtain an about 50-fold molar excess and incubated at room temperature (RT) for 2h. The biotinylation was stopped by dialysis against PBS. Aliquots of 50 μL were prepared and kept frozen at -20 ℃. A biotinylation degree of 4.7 mol/mol IgG was determined for this preparation. Although not checked explicitly, there was no evidence for substantial changes in avidity and selectivity compared with the native antibody.
Elisa development studies
Different assay conditions were investigated. In particular, the performance of the affinity-purified 70 R 10598 and the "normal" 20-R1379 rabbit anti-FXI antibody was compared side-by-side using the two preparations as a capturing antibody together with the biotinylated detection antibody. Standard coating conditions (10 μg/mL, pH 9.5) and different coating concentrations (1-20 μg/mL) and two pH values of the coating solution (pH 7.2 vs. pH 9.5) were investigated. Furthermore, the possible influence of benzamidine, usually added to the dilution buffer (DB), was investigated. The specificity of the paired antibodies was checked with human FXI-deficient/depleted plasma preparations and by using commercially available purified FXI/FXIa preparations.
Description of the ELISA
For the ELISA, the following buffers were used: Coating buffer (0.1 M Na2CO3, 0.1 M NaHCO3, pH 9.5.); washing buffer PBST (phosphate-buffer saline with Tween 20; 0.8% NaCl, 0.02% KCl, 0.02% KH2PO4, 0.126% Na2HPO4 × 2H2O, 0.05% Tween 20, pH 7.2); DB (0.1% BSA with 2 mM benzamidine in PBST); stopping solution (1.5 M H2SO4). Briefly, 100 μL/well rabbit anti-human FXI IgG 20-R1379, diluted 1/500 to 10 μg/mL in coating buffer was incubated at +4 ℃ overnight. The plate was then washed with PBST; blocking was done with 200 μL DB/well at 37 ℃ for 60 min. After a washing step, 100 μL DB/well was added, before the pre-diluted standard, control or samples were serially diluted 1+1 directly on the plate. Wells B11 and B12 served as assay blanks, containing all reagents but dilution buffer instead of sample. The plate was incubated at RT for 60 min and washed afterwards. Then, 100 μL/well of biotinylated anti-FXI antibody, diluted 1/1,000 in DB, was incubated at RT for 60 min. The incubation was terminated by a washing step. Streptavidin peroxidase (100 μL/well), diluted 1/4,000 in DB, was then added and incubated at RT for 30 min. A washing step terminated this final incubation. SureBlue was added and incubated at RT for 10 min; the color reaction was stopped with 1.5 M sulfuric acid (100 μL/well in both cases). The plate was measured at 450 nm (reference wavelength 620 nm). The calibration curve was constructed on each plate. It ranged from 0.07 to 2.30 mU FXI/mL (corresponding to 0.35 to 11.5 ng/mL) and was obtained by log-log fitting of the blank-corrected optical densities (ODs) and the FXI concentrations of the six calibrators D1 to D6. Samples were measured in duplicate serial dilution series. Only ODs within the calibration curve range were interpolated and the dilution corrected results were averaged to yield the final result. If required for the transformation of FXI plasma units to mass, an FXI plasma concentration of 5 μg/mL was used [3].
Setup of the assay validation
The assay attributes accuracy, precision, linearity, specificity, and lower limit of quantification were addressed. Three approaches were used for describing the ELISA's accuracy: Assessment of the assay calibration curves by using the back-fitting approach, spike-recovery in all three IgG products and comparison of the FXI concentration determined for the ISTH/SSC#4 preparation with its labelled FXI concentration. Precision was addressed on the level of intra-run (repeatability) and inter-run precision (intermediate precision). In particular, the inter-run precision was determined for the four process intermediates and all three IgG products, while intra-run precision was determined for the three final products only. Linearity of response was checked not only for the assay's calibration curves and dilution series of the IgG products with and without spiked FXI but also by a spike-recovery study carried out in FXI-deficient plasma using purified FXI and FXIa at concentrations from 0.05 to about 10 U/mL. The assay's lower limit of quantitation was confirmed for the matrix of all three IgG products by spiking 2.3 mU/mL FXI into the samples' dilutions and determining FXI recovery. Assay specificity was confirmed by the measuring FXI-deficient plasma. All calculations were done with Microsoft Excel for Microsoft 365 MSO.
Results
Results for the commercial Assaypro ELISA kit
Two runs were carried out using the commercial FXI ELISA kit Assaypro. In both runs, a purified FXI preparation provided acceptable dilution-response curves within the FXI concentration range of 5.9 to 95 ng/mL. Unexpectedly, two human reference plasma preparations provided clearly higher responses: Thus, the FXI concentration of 5 ng/mL in the plasma matrix yielded a blank-corrected OD of about 0.4, while purified FXI at 6 ng/mL provided an OD of only 0.031. This finding suggested a selectivity deficit resulting in a false positive response despite the high starting dilution of 1/1,000 that was applied for analysis of the plasma samples. In addition, the dilution-response curves of the human plasma preparations showed slopes clearly lower than that determined for the purified FXI preparation. Finally, a GGL sample with a known FXI level of about 50 mU/mL did not show any reactions. These data triggered the development of our FXI ELISA.
Results of the assay development
Comparison of a "normal" and an affinity purified anti-FXI IgG preparation: Polyclonal anti-FXI antibody preparations are commercially available from Fitzgerald at two qualities: "normal" and affinity purified. We compared the performance of both preparations. Figure 1 shows the calibration curves, obtained for the reference plasma preparation. These curves did not show any difference between the two anti-FXI preparations as the curves had almost identical slopes and showed good log-log curve fitting, demonstrated by the correlation coefficients, the relative total errors (RTEs) and the agreement of the back-fitted concentrations to their respective nominal ones. Moreover, there was no difference in the performance observed for the GGL matrix, characterized by an at least 10-times higher IgG level than that present in plasma and acidic pH. The dose-response curves obtained for the GGL sample, although starting at the low dilution of 1/20, did not evidence any matrix impact on the assay performance, specifically expressed as parallelism and linearity, and compared to that of the plasma dose-response curves. Furthermore, also the slopes obtained for the purified FXI preparation were very close to that obtained for the plasma sample. We therefore concluded that FXI capturing took place at the high selectivity required given the low abundance of FXI in human plasma. With regard to the two anti-FXI preparations, however, this analysis showed neither of the two preparations, tested head-to-head, was superior. Therefore, the preparation requiring less preparative efforts was selected.
Differentiation between FXI and activated FXI: The reasonable assumption that a polyclonal antibody preparation cannot differentiate the amino-acid-sequence-identical molecules FXI and FXIa was verified. FXI-deficient/depleted plasma was spiked with FXI or FXIa. Figure 2 shows the resulting dilution-response curves. The FXI-deficient and the FXI-depleted plasma, measured at the dilution 1/10, showed mean blank-corrected ODs of 0.008 and 0.010, respectively. These data confirmed the specificity of the FXI antibody. Spiked FXI and FXIa were selectively captured from the complex matrix of FXI-deficient/depleted plasma. The slope determined for the dilution-response curve of FXIa in buffer was 101.1% of that obtained for the plasma sample. This result verified the assumption that FXIa and FXI cannot be differentiated by the ELISA. The parallelism of the dilution-response curves found for FXI-/FXIa-spiked FXI-deficient/depleted plasma samples was acceptable as the slopes obtained differed by less than 11% from those of the respective plasma dilution series. Recoveries of 102.5% and 104.8% and of 94.3% and 96.2% were found for FXI and FXIa, spiked to the FXI-deficient and the FXI-depleted plasma, respectively. These data confirmed the reasonable assumption that the polyclonal anti-FXI antibody will not be able to differentiate FXI and FXIa.
Coating concentration: Coating of the capturing antibody is an important step which can determine assay performance. Apart from antibody concentration, also the pH is known to play an important role. The coating conditions were varied for the pH of the coating solution (pH 9.5 vs. pH 7.2) and the antibody concentration using coating concentrations from 1 to 20 μg/mL. Table 1 summarizes the results of this study, which was primarily evaluated with regard to the dose-response curves obtained for the reference plasma preparation diluted to FXI concentrations ranging from 4.6 to 0.14 mU/mL. Similar dose-response curves were obtained under all conditions tested. Only slight OD differences were found for the different coating concentrations, with slightly lower values obtained for the PBS at pH 7.2. Plausibly, the blank to D1 ratio increased with the coating concentration but reached a maximum of 1.69% at the highest coating concentration (20 μg/mL) in PBS. This ratio was considered still acceptable and was expected not to influence the assay performance. The dose-response curve attributes slope, correlation coefficient r and relative total error RTE were also similar and did not clearly favor any of the conditions tested. This was also true for the sensitivity variable OD per mU FXI, with slightly higher values found at pH 9.5. In summary, the data demonstrated the robustness of the coating process as changes in the coating concentration from 1 to 20 μg/mL were shown to have no substantial influence on the signal intensity or the quality attributes of the dose-response curves. Coating at pH 9.5 was shown to provide moderately better outcomes and was, therefore, selected for the final method.
Influence of benzamidine: Proteolytic degradation of antigen during the assay could affect assay performance especially when analyzing plasma protein matrices. Addition of benzamidine acting as a non-specific protease inhibitor is supposed to reduce this risk. We confirmed that the addition of benzamidine to the dilution buffer had no influence on the ELISA. Benzamidine was added to the BSA dilution buffer at a concentration of 2 mM to serve as a non-specific protease inhibitor. The calibration curves ranged from 2.30 to 0.07 mU/mL. Table 2 shows the influence of benzamidine on the calibration curve, presenting the relevant calibration curve attributes slope, correlation coefficient and RTE, the sensitivity measure OD/mU FXI and the FXI concentrations determined for a GGL sample. The two calibration curves were almost indistinguishable in all attributes and overlapped. Benzamidine had no influence on the sensitivity of the assay, as shown by the attribute OD/mU FXI which was 0.565 and 0.558 for the curves with and without benzamidine, respectively. The curve fitting was almost perfect with correlation coefficients of at least 0.9995 for the six-point calibration curves and RTEs not exceeding 8.3%. The FXI concentrations determined for a GGL sample were also almost identical as 0.068 and 0.067 U/mL were determined with and without benzamidine, respectively. In summary, the data demonstrated that benzamidine within the range investigated had no impact on the FXI ELISA performance.
Assay validation data
Results of the accuracy analysis: Figure 3 shows the mean six-point calibration curve (n = 40), obtained by log-log fitting. The curves ranged from 0.07 to 2.3 mU FXI/mL (0.35 to 11.5 ng/mL). The mean blank-corrected ODs of the six assay calibration standards had RSDs ranging from 11.0% to 17.9%, while the mean assay blank showed an RSD of 17.0%. The resulting calibration curves were similar in shape, i.e., parallel, as indicated by the RSD of 4.4%, determined for their mean slope. Also, the linearity of the curves was good, as shown by the mean correlation coefficient of 0.9997 (range: 0.9989 to 1.0000) and the very low mean RTE of 5.8% (range: 1.6% to 12.1%). With respect to the quality attribute RTE, it is worth mentioning that only two out of 40 RTEs were higher than 10%. This confirmed the high accuracy of the calibration curve fitting applied for the ELISA. In line with these data, the mean agreement between the nominal and the back-fitted concentrations of the six assay calibrators was within a 100 ± 3% range for all six concentrations of the calibration curve. Moreover, all individual back-fitted values were within a 100 ± 9% range. These data demonstrated the good accuracy and reproducibility and, consequently, the suitability of the model defined for the construction of the six-point calibration curves.
Furthermore, accuracy was more specifically determined by spike-recovery experiments carried out for the three relevant IgG products, GAMMAGARD LIQUID, GAMMAGARD S/D and CUVITRU. These products were spiked 1+1 with 4.60 mU FXI/mL to obtain their respective minimum sample dilution, i.e., 1/100 for GGL and CUV and 1/40 for GG-SD. This procedure enabled us to detect any detrimental influence caused by the samples' high human IgG concentration. Table 3 shows the recoveries of spiked FXI obtained in six separate runs done by three operators. The recovery of FXI spiked to the GGL sample ranged from 94.9% to 103.3%, resulting in the mean recovery of 99.1%, while the recoveries determined for the GG SD and CUV samples ranged from 93.4% to 102.9% and 105.6% to 109.4%, resulting in the mean recoveries of 99.4% and 106.8%, respectively. These data also met the acceptance criteria of 100 ± 20% defined for ligand-binding assays by the guideline for bioanalytical assay validation [27]. In summary, these data also confirmed the accuracy of the FXI ELISA in the sample matrix of IgG preparations, characterized by high IgG levels and acidic pH values.
In a separate approach, measurement of the international reference plasma ISTH/SSC#4 served to confirm the accuracy of the assay calibrator used. This reference preparation has a labelled FXI concentration of 0.89 U/mL, determined on the basis of the 1st WHO standard 04/102 for FXI [28]. As the anti-FXI antibody was shown not to discriminate between FXI and FXIa and provided similar ELISA responses for both molecules, the WHO standard 11/236 for FXIa was measured as well. This standard has an arbitrarily assigned FXIa potency of 10 IU/mL [23] which is not related in any way to the FXI plasma concentration (Elaine Gray, personal communication, 2015). Table 4 shows the FXI concentrations obtained for the ISTH/SSC#4 reference plasma in six independent measurements, done by three operators and the FXIa concentration, determined for the WHO standard in six measurements in one run. The agreement of the concentrations found with the respective labelled concentration is shown for both standards. The mean FXI (mean ± SD) concentration was 0.88 ± 0.013 U/mL, which represented a mean agreement of 98.9% to the labelled FXI concentration of the ISTH/SSC#4 standard. All individual recoveries differed by not more than 2.5% from the labelled concentration. These data confirmed the accuracy of the FXI concentration assigned to the reference plasma used for the assay calibration. The mean concentration determined for the WHO FXIa standard was 0.30 ± 0.008 U/mL. This represented only 3% of the standard's labelled FXIa concentration and showed that the arbitrarily defined international units (IUs) for FXIa do not absolutely correspond with FXI plasma units. Based on these data obtained with this particular ELISA, one IU of FXIa as defined by the WHO standard 11/236, corresponds to 0.03 plasma units of FXI.
Results of the precision analysis: Intra-run and inter-run precision were determined for four process intermediates and the three IgG products. Table 5 summarizes the data obtained by six measurements, done on one plate for the intra-run and in six independent runs done by three operators for inter-run precision, respectively. Intra-run precision was determined for the IgG products only. Overall, none of the RSDs determined exceeded 9.5%. As expected, slightly higher RSDs were found for the means of the independent measurements. The inter-assay precision determined for IgG products and intermediates did not differ obviously, despite clear differences in the FXI levels of the intermediates, ranging from 19.5 to 950 mU/mL. In particular, the intra- and inter-assay precision for the IgG products, expressed as RSDs, ranged from 3.9% to 6.2% and 4.1% to 9.2%, respectively. The precision data obtained during the assay validation confirmed the good performance of the FXI ELISA in all relevant matrices. Figure 4 completes the precision analysis by showing data obtained over an extended period: In particular, the FXI concentrations determined for a GGL sample in 15 independent runs are shown together with the data of the assay control. This lyophilized plasma sample was measured 83 times over 2 years. RSDs of 5.6% and 5.7%, for the GGL sample and the assay control, respectively, confirmed the good assay precision over the extended time. All data obtained were normally distributed. In addition, almost flat regression lines with coefficients of determination R² < 0.05 strongly suggested the stability of the ELISA over time.
Linearity analysis: The ELISA's design purposely facilitates the assessment of the parallelism of the samples' dilution-response curves with the corresponding calibration curve. The RSDs of the mean FXI concentrations, obtained for the individual dilution-corrected concentrations of the serial dilution series, are used for this parallelism assessment. According to published data, an RSD of less than 15% demonstrates adequate parallelism of the dilution series [29]. This criterion has been established for actual sample testing to guarantee adequate parallelism of the samples' dose response curves with that of the calibration curve. In addition, the parallelism of the dose-response curves obtained for the three IgG products analyzed at their respective minimum dilution with and without spiked FXI was investigated. Figure 5 shows the mean (n = 6) dilution-response curves and Table 6 the corresponding numerical data of these curves. The mean concentration-response curves obtained for the neat and FXI-spiked samples were linear as shown by their absolute correlation coefficients that were at least 0.9994 for all dilution series. The slopes of the dilution-response curves were similar between the assay standard and IgG samples with differences of less than 6.5% between the two slopes. Similar data, however, were obtained at even lower dilutions of 1/10, 1/20 and 1/40 of GG-SD, GGL and CUV, respectively, when IgG concentrations of up to 5 mg/mL were loaded to the plate (data on file). Overall, the data showed the linearity and the parallelism of the dose-response curves, confirming that the specific sample matrix of IgG products had no influence on the assay performance. Next, we checked assay linearity by adding defined amounts of purified FXI and FXIa to FXI-deficient plasma, thus obtaining FXI/FXIa concentrations ranging from 0.05 up to about 10 U/mL. Figure 6 shows the linear regression curve between the mean FXI/FXIa concentrations measured and their nominal concentrations. These regression curves showed correlation coefficients of 0.9999 and 1.0000 for FXI and FXIa, respectively. The slopes of the two curves were close to 1, with slopes of 1.0102 and 0.9494 found for FXI and FXIa, respectively. The data confirmed the linearity of the FXI ELISA within the range of 0.05 up to about 10 U/mL for both FXI and FXIa.
Specificity analysis: ELISA specificity is mainly dependent on the specificity of the antibody combination applied, which is usually guaranteed by its supplier. To confirm the FXI ELISA specificity, a human FXI-deficient plasma preparation, containing less than 1% of residual FXI activity according to the manufacturer's specification, was measured in six runs. Table 7 shows the FXI concentrations obtained and blank-corrected ODs measured together with those of the corresponding assay calibrator D6 with an FXI concentration of 0.07 mU/mL. The FXI concentration measured for the FXI-deficient human plasma preparation was less than 1.4 mU/mL in six measurements. This clearly met the specification of less than 1% residual FXI activity set by the supplier of this biological preparation. The blank-corrected ODs found for the 1/20-dilution of the FXI-deficient plasma were clearly lower than those of the corresponding assay standards D6 with a FXI concentration of 0.07 mU/mL, counting for not more than 32.6%. These data confirmed the specificity of the antibodies applied for the immunological measurement of FXI. This antibody combination, however, is not specific for non-activated FXIa, but also binds to FXIa, as shown by the data obtained. Thus, the ELISA does not discriminate between FXI and FXIa.
Limit of quantification: The limit of quantification of 0.01 U/mL for all three IgG products, measured at their minimum sample dilution of 1/40 and 1/100 for GG-SD and GGL/CUV, respectively, was confirmed by repeated spiking-recovery of these samples. Mean recoveries (n = 6) within a 100 ± 7% range with corresponding RSDs of lower than 3.5% confirmed the limit of quantification of 0.01 U FXI/mL.
Discussion
Patients suffering from primary antibody deficiencies require lifelong IgG replacement therapy [30,31], which was successfully established more than 30 years ago. This replacement therapy is based on intravenous, subcutaneous [32] or recombinant hyaluronidase-facilitated [33] administration of human polyclonal IgG, purified from pooled human plasma. The therapy's goal is to reduce the incidence and severity of infections and prevent long-term deterioration of organ function. Usually, this requires normalization of serum IgG levels, and for the intravenous route most patients are treated with IgG doses of 400 mg/kg body weight every 4 weeks. According to data presented at the European Blood Alliance and International Plasma and Fractionation Association in Amsterdam by the Marketing Research Bureau [34] the demand for polyclonal IgG was about 107 metric tons in 2010. Given this demand and the importance of IgG replacement therapy for primary antibody-deficient patients, the sudden increase in thrombotic events observed in 2010 caused by specific IVIG and SCIG brands prompted joint agency and manufacturer efforts to identify the root cause of these events. Ultimately, their higher incidence could primarily be associated with IgG preparations demonstrating low residual amounts of activated human coagulation factor XI. A panel of assays was set up for measuring FXIa in the matrix of IgG preparations. These included coagulation-based non-activated partial thromboplastin time and thrombin generation assays and more or less selective chromogenic assays. Assay development was completed when international FXIa reference preparations were established [23-25]. Their continued use will definitely harmonize and increase common quantitative understanding of FXIa levels in IgG products.
While analytical efforts almost exclusively focused on measuring FXIa activity, measurement of FXI protein (zymogen) was not generally considered as equally important. At first glance, this seems reasonable as FXIa activity ultimately triggers the thromboembolic events. Nevertheless, the level of FXI protein could be seen as essential additional information as it allows conclusions to be drawn regarding the maximum potential FXIa activity, given that FXI (auto)activation takes place in the matrix of an IgG preparation. Interestingly, such an event was described just recently, when increased FXIa levels were found in an IgG preparation 5 and 8 months after initial testing [22]. Data on FXI protein levels would not only have substantiated this finding but also have made it possible to predict if a further increase in FXIa could be expected or the maximum level had already been reached. The basis for such a prediction is provided here by the ELISA measurement of the WHO standard 11/236, where 1 IU of FXIa corresponded to 0.03 plasma units of FXI protein. This unexpected low ratio between the IU FXIa and plasma units of FXI, the latter established by reference to the 1st international standard 04/102 for FXI [28,35], is due to the arbitrary definition of the IU of FXIa. Its definition was not intended to reflect specific molecular alterations occurring during FXI activation [personal communication, Elain Gray, 2015]. In fact, the polyclonal anti-FXI IgG preparation used for the ELISA developed here does not differentiate between FXI and FXIa. The FXI activation process, triggered by activated factor XII, proceeds via cleavage of the single peptide bond between arginine 369 and isoleucine 370. The cleaved peptide chains are still kept together by disulfide bonds after activation but could of course undergo structural alterations as reflected by the serine protease activity obtained. These structural alterations, however, seem to be substantial enough to allow the development of FXIa-specific monoclonal antibodies, described recently [36].
For the ELISA described here, a single polyclonal anti-FXI antibody IgG preparation was used as supplied and after in-house biotinylation to obtain a detection antibody. This approach was based on the reasonable assumption that a polyclonal antibody preparation is expected to contain a variety of IgGs binding to different epitopes of FXI. The existence of the FXI homodimer made this approach even more promising as every possible epitope will be displayed not only twice but also most probably with some distance between both repetitions thus avoiding steric hindrance for IgG binding. The biotinylation of the anti-FXI IgG followed a standard protocol with a 50-fold molar excess of Sulfo-NHS-LC-Biotin. This activated reagent addresses the primary amines on the IgG preparation to be biotinylated. The biotinylation degree of 4.7 mol biotin/mol IgG obtained was within the usually obtained modification degrees for other successfully applied IgG preparations and did not obviously alter the binding features of the anti-FXI IgG. Interestingly, direct comparison of an affinity-purified FXI preparation with the non-purified antibody did not reveal any obvious benefits of the more highly purified preparation. Therefore, we decided to use the non-affinity purified anti-FXI IgG preparation because less effort was required to purify it and, therefore, the risk of denaturation associated with harsh elution conditions probably required during the affinity purification was lower. The specificity claimed by the manufacturer of the anti-FXI IgG was confirmed by testing patient derived FXI-deficient plasma and artificially FXI-depleted plasma. Both preparations elicited no measurable responses at low dilutions thus confirming the specificity of the antibody preparation for FXI. By contrast, the preparation was unable to differentiate between FXI and FXIa.
We developed the ELISA because we found the commercial FXI ELISA kit we used did not provide satisfactory data. In particular, we found unexpected differences between the slopes of the dilution-response curves obtained for plasma and purified FXI. In addition, no measurable response was obtained for an IgG preparation with a confirmed FXI level. These findings not only suggested a selectivity deficit, but also highlighted a special requirement associated with the analysis of samples characterized by high polyclonal IgG levels. A recent search using the web-based platform Biocompare identified six FXI ELISA systems. The assay sensitivities described for these commercially available ELISAs did not differ substantially from that of the ELISA presented here, but there were clear differences for the sample matrix: the use of the commercial ELISAs was described only for serum/plasma or buffer samples. Considering that plasma/serum samples will usually contain IgG levels of not higher than 10 mg/mL, the unique challenge of testing polyclonal IgG preparation with IgG levels of as high as 200 mg/mL becomes evident. As this testing requires adequate sensitivity, high sample dilution is inappropriate and high levels of human polyclonal IgG will be present in the diluted sample loaded to the plate. This could provoke interactions with the surface of the coated well. On the one hand, the coated antibody could be displaced from the well, which would most probably impact the assay performance. On the other hand, polyclonal IgGs could be the reason for non-specific binding reactions thus producing false-positive results. These sample-specific challenges associated with the ELISA measurement of IgG preparations can be addressed during assay development. Thus, our investigation of the antibody coating concentration demonstrated that coating at pH 9.5 provided moderately better outcomes than coating in PBS at pH 7.2. On the other hand, the data demonstrated the robustness of the coating process, as changes in the coating concentration from 1 to 20 μg/mL were shown to have no substantial influence on the signal intensity or the quality of the calibration curves. Similarly, the addition of 2 mM benzamidine to the sample dilution buffer had no impact on the assay performance.
Finally, the assay validation addressed the attributes accuracy, precision, linearity, and specificity, with a specific focus on the measurement of the three IgG preparations GAMMAGARD S/D, GAMMAGARD LIQUID and CUVITRU. In particular, accuracy was checked by assessing the calibration curves, measuring an international reference preparation for FXI and by spike-recovery for the three IgG products. The data obtained by these studies easily met the criteria defined by the guideline for bioanalytical method validation for ligand binding assays [27] although it has to remarked that such sample types are actually outside the scope of this guideline. Nevertheless, it at least addresses ligand-binding assays. Similarly, the precision profile determined for the IgG products was adequate and justified the ELISA's use for measuring FXI in the three IgG products and related process intermediates. Most importantly, however, the dilution-response curves obtained for the three IgG products did not provide any evidence for a detrimental influence of the high human IgG concentrations present in the tested samples. The regression curves calculated for the neat and FXI-spiked IgG preparations were highly parallel to the curve of the reference plasma preparation, used for the assay calibration.
In summary, we describe the development of a robust, sensitive and specific ELISA for the measurement of human FXI based on using a commercially available anti-FXI IgG antibody preparation, which is also used for the preparation of a biotinylated detection antibody. The data obtained by the assay validation qualified the method to be used for measuring FXI in intermediates and final IgG products. As the antibody combination used does not differentiate between FXI and FXIa, a relation between FXIa concentrations, expressed in IU/mL, and FXI concentrations, expressed in U/mL, could be established for the first time to the best of our knowledge. This relation will be useful for the predicting maximum levels of FXIa at given levels of FXI in an IgG product.
Conflict of Interest
All authors are full time employees of Baxalta Innovations GmbH, a Takeda company.
Acknowledgments
The authors thank Sonja Haindl, Sandra Hainzelmayer, Eva Minibeck, Gabriela Mohr, Hubert Ritter and Carina Zierfuss for skillful technical assistance; Susanne Brunner and Sabine Riedler are acknowledged for review of the experimental data. Editorial support was provided by Elise Langdon-Neuner, funded by Baxalta Innovations GmbH. The work was sponsored by Baxalta Innovations GmbH, a Takeda company.

