
International Journal of Analytical and Bioanalytical Methods
(ISSN: 2633-8912)
Volume 2, Issue 1
Review Article
DOI: 10.35840/ijabm/2408
Analysis of Total Amino Acids in Rice Using a Validated Reversed-Phase High Performance Liquid Chromatographic Method with Diode Array Detection (RP-HPLC-DAD)
GVV Liyanaarachchi1,2*, KRR Mahanama2, HPPS Somasiri1, PAN Punyasiri3 and JD Kottawa-Arachchi4
Table of Content
Figures
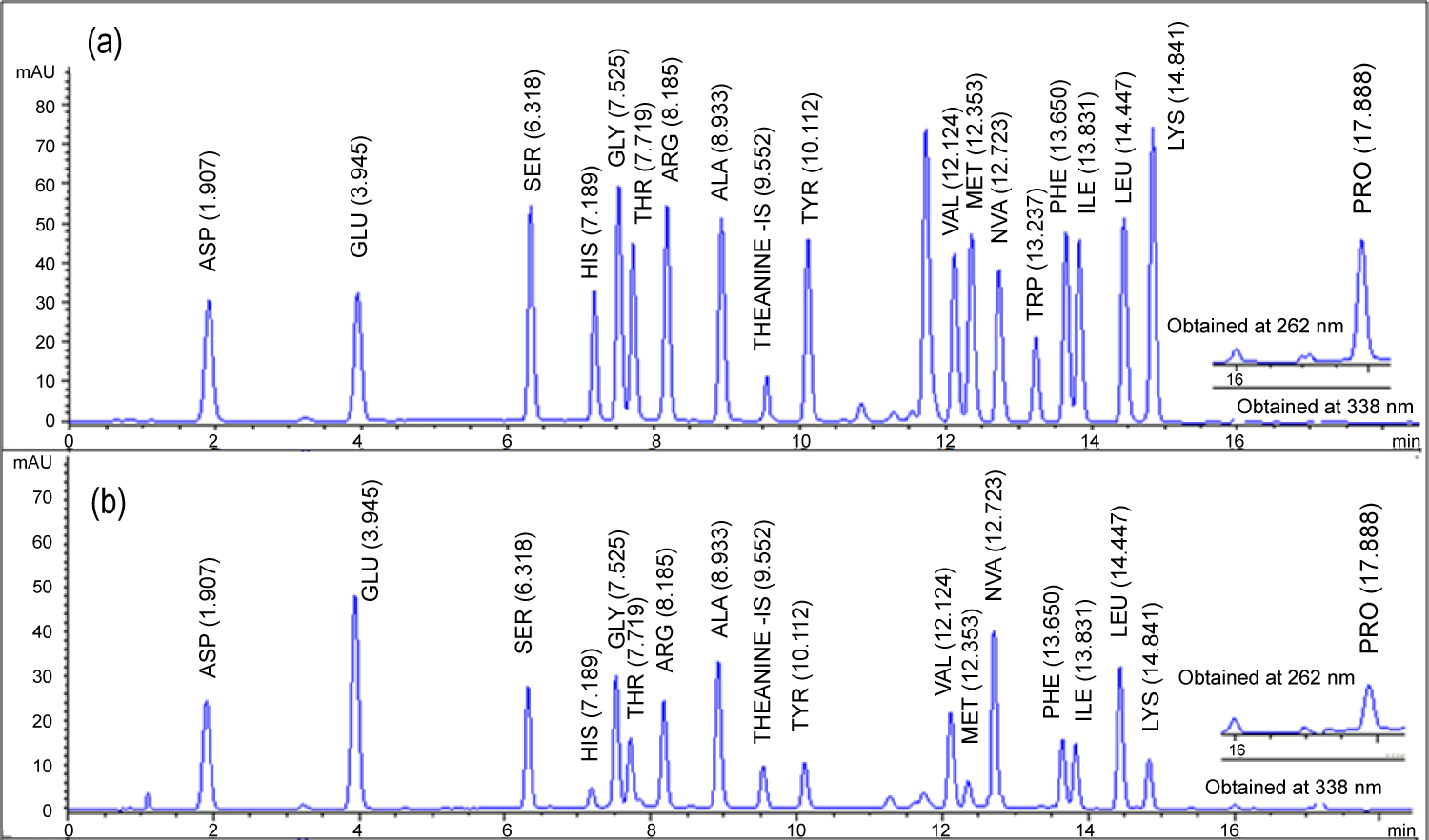
Figure 1: LC-MS/MS quantifier ion chromatograms...
LC-MS/MS quantifier ion chromatograms (sMRM mode) of a milk sample fortified at 0.5 MRL/RC of the studied analytes.
Tables
Table 1: Injector program.
Table 2: Accuracy of amino acid analysis.
Table 3: Precision, recovery and selectivity.
Table 4: Working range, Linearity, LOD, LOQ and Uncertainty of the method.
Table 5: Total amino acid contents of traditional rice cultivars grown in Batalagoda RRDC.
Table 6: Total amino acid contents of traditional rice cultivars grown in Bombuwala RRDC.
References
- SS Gnanamanickam (2009) Biological control of rice diseases: Rice and its importance to human life. Springer, USA.
- G Kennedy, B Burlingame (2003) Analysis of food composition data on rice from a plant genetic resources perspective. Food Chem 80: 589-596.
- JS Kamara, S Konishi, T Sasanuma, T Abe (2010) Variation in free amino acid profile among some rice (Oryza sativa L). Cultivars Breed Sci 60: 46-54.
- DK Verma, PP Srivastav (2017) Proximate composition, mineral content and fatty acids analyses of aromatic and non-aromatic indian rice. Rice Sci 24: 21-31.
- BG Lyon, ET Champagne, BT Vinyard, WR Windham, FE Barton, et al. (1999) Effects of degree of milling, drying condition, and final moisture content on sensory texture of cooked rice. Cereal Chem 76: 56-62.
- M Martin, MA Fitzgerald (2002) Proteins in rice grains influence cooking properties. J Cereal Sci 36: 285-294.
- L Xie, N Chen, B Duan, Z Zhu, X Liao (2008) Impact of proteins on pasting and cooking properties of waxy and non-waxy rice. J Cereal Sci 47: 372-379.
- (2005) Animal feeding stuffs - Determination of amino acids content. ISO 13903: 2005.
- M Fountoulakis, H Lahm (1998) Hydrolysis and amino acid composition analysis of proteins. J Chromatogra A 826: 109-134.
- SA Cevikkalp, GB Loker, M Yaman, B Amoutzopoulos (2016) A simplified HPLC method for determination of tryptophan in some cereals and legumes. Food Chem 193: 26-29.
- C Mota, M Santos, R Mauro, N Samman, A Sofia, et al. (2016) Protein content and amino acids profile of pseudocereals. Food Chem 193: 55-61.
- P Pal, P Kaur, N Singh, AP Kaur, NN Misra, et al. (2016) Effect of nonthermal plasma on physico-chemical, amino acid composition, pasting and protein characteristics of short and long grain rice flour. Food Res Int 81: 51-57.
- K Liu, J Zheng, F Chen (2017) Relationships between degree of milling and loss of Vitamin B, minerals, and change in amino acid composition of brown rice. LWT-Food Sci Technol 82: 429-436.
- I De Biase, A Liu, T Yuzyuk, N Longo, M Pasquali (2015) Quantitative amino acid analysis by liquid chromatography-tandem mass spectrometry: Implications for the diagnosis of argininosuccinic aciduria. Clin Chim Acta 442: 73-74.
- DE Otter (2012) Standardised methods for amino acid analysis of food. The British J Nutr 108: S230-S237.
- R Dave, RD Tripathi, S Dwivedi, P Tripathi, G Dixit, et al. (2013) Arsenate and arsenite exposure modulate antioxidants and amino acids in contrasting arsenic accumulating rice (Oryza sativa L) genotypes. J Hazard Mater 262: 1123-1131.
- H Ning, J Qiao, Z Liu, Z Lin, G Li, et al. (2010) Distribution of proteins and amino acids in milled and brown rice as affected by nitrogen fertilization and genotype. J Cereal Sci 52: 90-95.
- DJ Dietzen, AL Weindel, MO Carayannopoulos, M Landt, ET Normansell, et al. (2008) Rapid comprehensive amino acid analysis by liquid chromatography/tandem mass spectrometry: Comparison to cation exchange with post-column ninhydrin detection. Rapid Commun Mass Spectrom 22: 3481-3488.
- K Dettmer, AP Stevens, SR Fagerer, H Kaspar, PJ Oefner (2012) Amino acid analysis in physiological samples by gc-ms with propyl chloroformate derivatization and iTRAQ-LC-MS/MS. Methods Mol Biol 828: 165-181.
- WP Chen, XY Yang, AD Hegeman, WM Gray, JD Cohen (2010) Microscale analysis of amino acids using gas chromatography-mass spectrometry after methyl chloroformate derivatization. J Chromatogra B 878: 2199-2208.
- B Zhang, ZQ Rong, Y Shi, JG Wu, CH Shi (2011) Prediction of the amino acid composition in brown rice using different sample status by near-infrared reflectance spectroscopy. Food Chem 127: 275-281.
- SM Rutherfurd, BM Dunn (2011) Quantitative amino acid analysis. Curr Protoc Protein Sci 63: 1-6.
- HK Nielsen, RF Hurrell (1985) Tryptophan determination of food proteins by h.p.1.c. after alkaline hydrolysis. J Sci Food Agric 36: 893-907.
- A Rita, F Roberta, B Remo, R Mena, M Flavio (2009) Nutritional and physicochemical characterization of italian rice flours and starches. Food Sci Technol Res 15: 507-518.
- EJ Park, JW Chung, YJ Park, IM Chung, JK Ahn, et al. (2009) Association analysis of the amino acid contents in rice. J Integr Plant Biol 51: 1126-1137.
- F Tanaka, F Tanaka, A Tanaka, T Uchino (2017) Effect of high temperature drying on amino acid decomposition in feed rice. Eng Agric Environ Food 10: 1-3.
- J Xiao-ling, T Ji-chun, HA Zhi, Z Wei-dong (2008) Protein content and amino acid composition in grains of wheat-related species. Agric Sci China 7: 272-279.
- MS Khan, E Ali, S Ali, WM Khan, MA Sajjad, et al. (2014) Assessment of essential amino acids in wheat proteins: A case study. J Environ Biol 4: 185-189.
- L Vilmane, S Zute, E Straumîte, R Galoburda (2015) Protein, amino acid and gluten content in oat (Avena sativa L.) grown in Latvia. Proceedings of the Latvian academy of sciences, 69: 170-177.
- K Szkudzinska, I Smutniak, J Rubaj, W Korol, G Bielecka (2017) Method validation for determination of amino acids in feed by UPLC. Accredit Qual Assur 22: 247-252.
- (2012) FDA guidelines for the validation of chemical methods for the FDA foods program. US Food and Drug Administration.
- (2014) The fitness for purpose of analytical methods; A laboratory guide to method validation and related topics. Eurachem.
- (2016) Appendix F: Guidelines for standard method performance requirements. AOAC International.
- JW Henderson, RD Ricker, BA Bidlingmeyer, C Woodward (2000) Rapid, accurate, sensitive, and reproducible HPLC analysis of amino acids and the agilent 1100 HPLC. Agilent Technologies.
- R Sutarno, HF Steger (1985) The use of certified reference materials in the verification of analytical data and methods. Talanta 32: 439-445.
- P Ravisankar, CN Navya, D Pravallika, DN Sri (2015) A review on step-by-step analytical method validation. IOSR J Phar 5: 7-19.
- (2008) Evaluation of measurement data-guide to expression of uncertainty in measurement. JCGM 100, GUM 1995 with minor corrections.
- M Huang, H Zhang, C Zhao, G Chen, Y Zou (2019) Amino acid content in rice grains is affected by high temperature during the early grain-filling period. Sci Rep 9: 2700.
- J Wang, T Hasegawa, L Li, SK Lam, X Zhang, et al. (2018) Changes in grain protein and amino acids composition of wheat and rice under short-term increased [CO2] and temperature of canopy air in a paddy from east china. New Phytol 222: 726-734.
- S Oh, S Lee, S Park, S Lee, S Lee, et al. (2019) Statistical study on the environmental effects on the natural variation of nutritional components in rice varieties. Food Sci Nutr 7: 163-172.
Author Details
GVV Liyanaarachchi1,2*, KRR Mahanama2, HPPS Somasiri1, PAN Punyasiri3 and JD Kottawa-Arachchi4
1Industrial Technology Institute, Sri Lanka
2University of Colombo, Sri Lanka
3Institute of Biochemistry, Molecular Biology and Biotechnology, University of Colombo, Sri Lanka
4Tea Research Institute of Sri Lanka, Sri Lanka
Corresponding author
GVV Liyanaarachchi, Industrial Technology Institute, 363, Bauddhaloka Mawatha, Colombo 7; University of Colombo, Colombo 3, Sri Lanka.
Accepted: May 13, 2020 | Published Online: May 15, 2020
Citation: Liyanaarachchi GVV, Mahanama KRR, Somasiri HPPS, Punyasiri PAN, Kottawa-Arachchi JD (2020) Analysis of Total Amino Acids in Rice Using a Validated Reversed-Phase High Performance Liquid Chromatographic Method with Diode Array Detection (RP-HPLCDAD). Int J Analyt Bioanalyt Methods 2:008.
Copyright: © 2020 Liyanaarachchi GVV, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Adaptable to any routine laboratory, the study presents the validation results of a simple, accurate and reliable method developed for the analysis of total amino acids (TAAs) in rice using reversed-phase high-performance liquid chromatography-diode array detection (RP-HPLC-DAD).
Exhibiting excellent selectivity with resolution (Rs) ≥ 2 for seventeen amino acids, the method was proven accurate against the analysis performed on the certified reference material (CRM): NIST 3233. Percentages of recoveries were in the range 86%-100% with percentage relative standard deviation (%RSD) ≤ 6% for all amino acids. Limit of detection (LOD) and limit of quantification (LOQ) values were within 0.024-0.069 g/100 g and 0.025-0.078 g/100 g respectively. A wide working range with satisfactory linearity having regression coefficients ≥ 0.999 were reported for all the amino acids.
Complying with international guideline requirements, this validated method can be successfully applied for the determination of seventeen TAAs including all essential amino acids in rice.
Keywords
Rice, RP-HPLC- DAD, Total amino acids, Validation
Introduction
As the dietary staple of more than half of the world population [1], rice (Oryza sativa L.) is recognized as one of the most important cereal crops in the world. Rice significantly contributes to the daily nutritional requirement as the major source of energy supply and the protein intake of the Asian diet [2]. Rice contains an average protein content of 4.5-15.9% [2]. Owing to the changes in the cultivar, environmental conditions, breeding techniques, agricultural practices and postharvest conditions, significant variations in protein levels both qualitatively and quantitatively were observed in rice [3,4]. Protein is the second most abundant nutrient present in rice which is only next to carbohydrates. Protein plays a significant role in determining the nutritional quality, functional properties, texture, pasting capacity and the sensory characteristics of rice [5-7]. The determination of amino acid composition of rice becomes vital for defining the characteristics related to the protein quality of rice. Several methods for determination of amino acids in rice and other matrices have been discussed in literature [8-14]. In amino acid analysis, generally proteins are hydrolyzed using 6 mol/L HCl solution at the temperature of 110 ℃ [8,9,13,15] for extended hours of 24 h or at temperatures above 110 ℃ for periods shorter than 24 h [9,16,11]. The hydrolyzed amino acids are then analyzed using either high performance liquid chromatography with ultra violet/visible, diode array or fluorescence detection (HPLC-UV/VIS/DAD/FLD) [10-12,16], amino acid analyzers [13,17], liquid chromatography-tandem mass spectrometric detection (LC-MS/MS) [14,18,19] gas chromatography-mass spectrometric detection (GC-MS) [14,19,20] or near-infrared reflectance spectroscopy (NIRS) [21].
It has been reported that the standard hydrolysis conditions do not guarantee the extraction of all the amino acids, most often excluding sulfur containing amino acids and tryptophan [15]. The studies have further indicated that amino acids analyzed under standard hydrolysis conditions can lead to either underestimation or over estimation of the actual value affecting the reliability of the results produced [15,22]. For example, losses encountered in tryptophan during the acidic hydrolysis can be overcome by performing the alkaline hydrolysis during the tryptophan analysis [10,23]. Thus the need for accurate and reliable methods to determine amino acid composition in rice becomes extremely important in defining its protein characteristics. The characterization of optimum hydrolysis conditions that produce acceptable recoveries for individual amino acids in the matrix assures the generation of reliable results. Therefore, evaluation of the method performance characteristics becomes paramount for the generation of accurate results.
There are several studies carried out on analysis of amino acids in rice [13,17,24-26] or other cereals [27-29]. However, except for the study on pseudo-cereals by Mota, et al. 2016 [11] and work by Szkudzinska, et al. 2017 [30] on rye, none of the work described in recent studies outlines a comprehensive validation study aimed at the evaluation of the method performance characteristics in analyzing the profile of amino acids in rice or cereal matrices which includes the complete profile of essential amino acids. The method developed by Mota, et al. 2016 [11] has excluded tryptophan analysis, hence the complete essential amino acid composition of the cereal matrix was not covered. Further, in their work, the evaluation of method performance characteristics has been performed based on the analysis of an ephedra containing protein powder which is a non-cereal, hence challenges the validity of the performance evaluation criteria. On the other hand, as done by Szkudzinska, et al. 2017 [30], performance of the method evaluated based on fortification of amino acids in free form does not guarantee the actual recoveries that would be obtainable for the protein bound amino acids. Most often sulfur containing amino acid analysis, involves an oxidation step of 16 h prior to hydrolysis [8,9,30].
Primarily, due to these complexities involved with the hydrolysis and the unavailability of a simple and reliable protein hydrolysis method, amino acid analysis often remains unattempted by most of the analytical laboratories in the routine framework. In this context, the aim of this study is to develop and validate a simple, accurate and reliable method that enables analysis of amino acid composition including all essential amino acids in rice with the use of the conventional laboratory oven and the RP-HPLC-DAD detection making the analysis simple and accessible to any routine analytical laboratory.
Methodology
Materials and methods
Chemicals and reagents: The standard reference materials of amino acids; L-aspartic acid (Asp), L-serine (Ser), L-glutamic acid (Glu), L-lysine (Lys), L-proline (Pro) glycine (Gly), L-histidine (His), L-arginine (Arg), L-threonine (Thr), L-alanine (Ala), L-tyrosine (Tyr), L-valine (Val), L-methionine (Met), L-isoleucine (Ile), L-leucine (Leu) and L-phenylalanine (Phe) (Sigma Alrich, Chemicals, St. Louis, MO), each of purity > 98% were prepared in 0.1 M HCl solution. Due to the limited stability in acidic solutions, L-tryptophan (Trp), L-theanine and L-norvaline (Nva) were prepared in ultra pure water and were stored for only two weeks. The certified reference material (CRM): NIST 3233 which was a fortified breakfast cereal was purchased from the National Institute of Standards and Technology (NIST), USA. L-Theanine (Baxter Smith Labs, USA), and L-Norvaline (Sigma Alrich, Chemicals, St. Louis, MO) with purity > 98% were used as the internal standards (ISs).
The derivatization of the primary amino acids was carried out using the o-phthalaldehyde 3-mercaptopropionic acid (OPA-MPA) as the derivatizing agent, while the secondary amino acid: Pro was derivatized using the 9-Fluorenylmethoxycarbonyl chloride (FMOC) (Agilent Technologies, USA). Other chemicals which were of analytical reagent grade and the HPLC grade solvents purchased from Sigma Aldrich were used for the preparation of samples and the two mobile phases which consisted of A(40 mmol/L Na2HPO4 with pH adjusted to 7.8 using a 10 mol/L sodium hydroxide solution) and B (45% acetonitrile, 45% methanol, 10% water).
Grain samples: The applicability of the method was assessed [31-33] using seven traditional rice varieties obtained from the regional rice research and development centers (RRRDCs) located at Batalagoda and Bombuwala in Sri Lanka.
Sample preparation
The unpolished rice samples after finely grinding using a laboratory grinder (IKA-MF 10 basic Microfine grinder drive), were sieved through a 0.3 mm sieve prior to analysis. To 0.2 g of the sieved sample placed inside a screw capped glass tube, 5.00 mL of the hydrolysis mixture (6 mol/L HCl containing 1% (v/v) thiodiglycol and 1 g of phenol per liter) was added [8] and vortexed for 5 minutes. The vortexed sample was placed inside a drying oven set at 110 ℃. During the first hour, in order to prevent a building up of pressure (due to the evolution of gaseous substances) and to avoid explosion, the screw cap was placed over the top of the glass tube without tightening. After 1 h, the glass tube was closed and left in the oven for 22 h. On completion of the hydrolysis, the glass tube was removed from the oven and once the mixture reaches the room temperature was carefully opened inside an ice-water bath. Initially, the pH of the hydrolyzed mixture was adjusted to around pH to 3 using a 10 mol/L sodium hydroxide solution while making sure that the temperature of the solution was kept below 40 ℃. The final adjustment of pH to 2.2 was carried out using a 1 mol/L sodium hydroxide solution. Finally the pH adjusted solution was transferred to a 25 mL volumetric flask and after addition of 100 µL of 50 nmol/L of both IS’s L-Nva and L-theanine, the resulting solution was made up to the mark with ultra pure water acidified to pH 2.2 with a 0.01 mol/L HCl solution.
Since Trp is destroyed during the acid hydrolysis, alkaline hydrolysis was performed using a 4.2 mol/L NaOH solution containing 1% (v/v) thiodiglycol for 18 h instead of the acid hydrolysis mixture mentioned above. The alkaline mixture resulting from the hydrolysis was adjusted to pH 2.2 with the use of 6 mol/L HCl solution finally being made up to 25 mL with ultra pure water acidified to pH 2.2 with a 0.01 mol/L HCl solution.
The prepared samples, after filtering through 0.45 µm syringe filter, were injected to the HPLC using automated pre-column derivatization.
HPLC analysis
The analysis was performed using an Agilent 1100 HPLC systems (Agilent Technologies, Palo Alto, CA) which consisted of a diode array detector (DAD) (G1321A). Pre-column online derivatization was achieved using the Agilent programmable auto sampler (G1313A). An Agilent Zorbax Eclipse AAA column with dimensions of (4.6 mm × 150 mm, 5 µm) was used for the chromatographic separation. The gradient elution started with 100% A for 1.9 min; ramped to 57% B within next 18.1 min; ramped to 100% B in 18.6 min and kept at 100% B till 22.3 min; then ramped to 100% A in 23.2 min and kept with 100% A till 26 min. The column was operated at 40 ℃ and the flow rate of the method was set at 2 mL/min throughout the runtime [34]. The Zorbax AAA guard columns (4.6 mm × 12.5 mm) were used to prolong the duration of the analytical column.
With the aid of the programmable autosampler and the injector program outlined in Table 1, the automated pre-column online derivatization with OPA-MPA and FMOC was performed prior to the injection of samples to the HPLC [34]. The derivatized primary amino acids were monitored at 338 nm while the secondary amino acid: Pro was monitored at 262 nm using the DAD detector. Agilent Chemstation software version B.04.03 was used for data acquisition and analysis.
Validation of the method
Validation method was carried out in compliance with the requirements specified in the international method validation guidelines [31-33]. The performance characteristics of the method in terms of accuracy, precision, recovery, selectivity, linearity, limit of detection (LOD), and limit of quantification (LOQ) were studied and the applicability of the method was tested on seven local traditional rice varieties.
The accuracy, precision, recovery and linearity of the method were evaluated based on the results obtained after the analysis of the certified reference material (CRM): NIST 3233 on breakfast cereals in six replicates.
In the absence of a matrix material free of amino acids, the determination of LOD and LOQ values were performed using six replicate analysis performed on the blank samples fortified at lowest quantifiable limits.
Statistical analysis
The statistical analysis was performed using the statistical software package, SAS for Windows V 9.1 (SAS Institute Inc., NC, USA). The level of significance was p < 0.05. Based on the percentage relative standard deviations (%RSD), intra and inter day precision and recovery values were evaluated. The estimation of the expanded uncertainty was evaluated at 95% confidence level including the factors contributing from repeatability, reproducibility and regression. The TAAs observed for the rice cultivars reported at the two locations were statistically analyzed using ANOVA and Duncan multiple range test.
Results and Discussion
The summary of the validation data in terms of accuracy, selectivity, precision, recovery, limit of detection (LOD), limit of determination (LOQ), linearity and measurement uncertainty obtained for the studied amino acids are listed in Table 2, Table 3 and Table 4.
Accuracy
The accuracy was evaluated based on the recovery values obtained for the certified reference material (CRM): NIST 3233 on breakfast cereals. The acceptance criterion for accuracy was calculated as per the equation 1 where : Assigned value of the certified reference material (CRM), : Mean value obtained for the CRM, : Uncertainty associated with the certified reference value and : Uncertainty associated with the analytical method for the particular analyte respectively. As summarized in Table 2, based on the criterion [35], the method was found accurate for analysis of all the studied amino acids.
Since an assign values for Pro was not available in the CRM, accuracy of the analysis was assessed based on the recovery values obtained for the fortified Pro at the middle of the working range.
Acceptance criterion: - (1)
Recovery
Recoveries of the amino acids were calculated based on the assigned values mentioned for the certified reference material (CRM): NIST 3233 on breakfast cereals. Since a value was not assigned for Pro in the CRM, recovery of Pro was calculated based on the fortification of Pro to the rice matrix at the mid level of the working range.
During the acid hydrolysis, Trp is entirely destroyed while Gln and Asn are completely oxidized to Glu and Asp respectively. However, inclusion of 1% (w/v) phenol as a protective agent in the hydrolysis mixture significantly reduced the loss of recoveries of the sensitive amino acids such as Met, Ser, Thr and Tyr [9]. Base hydrolysis aided optimum recovery for Trp which is 98%. Therefore, the overall mean recoveries obtained for the studied amino acids as given in the Table 3 were in the range 86%-100%. Except for Tyr, Val, Lys, Pro, Arg and Met, the recoveries for rest of the amino acids were within the accepted values for recovery for the specified analyte concentration levels recommended by the FDA Guidelines for the validations of Chemical Methods for the Foods Program [31].
Selectivity
The selectivity was assured based on the relative retention times calculated with reference to the respective IS obtained for amino acids in the sample to those obtained for amino acid reference standards. For polar amino acids, theanine was considered as the IS while non polar amino acids were normalized against the IS: Norvaline.
A minimal difference of ± 0.1% of relative retention times obtained for the amino acids in spiked matrices against the reference standards injected in blank solutions were considered acceptable. The resolution factors (Rs) calculated for all the amino acids were greater than 1 with the lowest Rs value of 2 obtained for Gly/Thr and Phe/Ile pairs (Table 3), where generally Rs ≥ 2 is regarded adequate [36]. Signifying excellent resolution which indicates better selectivity for all the analytes of interest as given in the Figure 1.
Precision
The precision of the method was measured under repeatable conditions on six replicate analyses of the CRM on the same day while the intermediate precision was calculated based on six replicate analysis carried out on the CRM sample by different analysts on different days over an extended period of three months.
The intermediate precision in analysis of each amino acid in the method (%RSD) was ≤ 6% (Table 4). These RSD values are well in compliance with the recommended RSD for the particular analytical range which is 6% as specified in the AOAC guideline on Method validation [33].
Limit of detection and limit of quantification
The LODs and the LOQs were calculated considering the mean and the standard deviation (SD) of the values obtained for the analyzed blank samples fortified at the lowest detection levels. The values were calculated by adding to the mean value, 3 times of the SD for LOD while 5 times of the SD values for LOQ respectively [32]. The LODs and the LOQs for the method were in the range 0.024-0.069 g/100 g and 0.025-0.078 g/100 g respectively allowing high sensitivity in detection of amino acids in lower levels as given in Table 4. The method reported lowest and highest LODs and LOQs for Gly and Tyr respectively. Miniaturization of the final total volume of the neutralized sample after hydrolysis aided achieving of low LOD and LOQ values allowing quantification of amino acids in rice and other cereals present in low levels.
Linearity and the working range
The calibration range consisted of seven calibration levels at 25, 50, 100, 250, 500, 1000 and 2000 μmol/L. The linear regression line was constructed using the peak area ratio of standard to IS at each concentration level. The regression coefficients obtained for all amino acids analyzed (R2) were ≥ 0.999 Hence, excellent linearity is demonstrated in the method over a wide working range as given in the Table 4. This enables the analysis of amino acids levels which are generally present in rice and other cereals in different levels in a single run.
Measurement uncertainty
The measurement uncertainty for each amino acid was calculated by considering the uncertainty contribution arising from repeatability, regression, standard preparation and sample preparation of the method [37].
The percentage expanded uncertainties for each amino acid, with a coverage factor of 2 (k = 2) were ≤ 7% for all the amino acids analyzed as given in Table 4. Except for Val and Met, Szkudzinska, et al. 2017 [30] reports uncertainties of similar magnitude in their method on amino acid analysis.
The uncertainties associated with the repeatability of the method and the regression analysis involved in the calibration step mainly constituted the percentage uncertainty, while the uncertainty associated with the preparation of the calibration standards and preparation of samples made up the remainder.
Amino acid composition in rice samples
The Table 5 and Table 6 summarize the composition of TAAs determined using the validated method in seven rice varieties cultivated in two locations in the country. As per the composition, Glu was the major amino acid present while Asp, Ala, Val, Gly and Leu levels also found in comparatively higher levels. Further, compared to the other analyzed amino acids, Trp, Met and His were found in relatively lesser quantities. In literature, several studies report similar compositions of Glu, Asp and Val in rice [13,17,24,25]. However, in comparison to those studies, relatively higher levels of Ala and Gly have been reported in the local traditional varieties.
When these data were statistically investigated using the Duncan Multiple Range Test it was revealed that there were significant variations (p < 0.05) in the individual amino acids and the mean total amino acid levels among the cultivars as well as between the two locations. The results were further analyzed to evaluate the impact of cultivar and location on the individual and total amino acid levels. Based on the analysis, it was revealed that there were significant variations in Asn, Glu, Thr, Arg, Ala, Tyr, Phe, Ile, Leu, Lys, Pro as well as mean total amino acid levels among the studied cultivars. Except for Gly, Thr, Arg, Ala, Phe and Ile, rest of the amino acids significantly varied between the two locations. Except for His, Tyr, Met and Pro, the interaction between cultivar (c) and the location (l), (c × l) was found significant for all the investigated amino acids, emphasizing the significance of impact of variety and the geographical conditions on the amino acid composition of rice. Studies have revealed that, with increasing temperatures of the environment, notable decrease in protein yield and amino acid content in rice [38] and protein content in wheat [39] have been reported. In addition, similarly as observed with this study, Oh, et al. (2019) [40] has pointed out that the amino acid composition is significantly affected by the rice variety and the location of cultivation. Therefore, these findings emphasize the significance of further investigations in this direction involving harmonization of genetic facts and agro climatic practices in breeding techniques for development of rice crops with optimum nutritional quality.
Conclusion
The study presents the validation data of a quantitative method for analysis of amino acids in rice using reversed phase HPLC with diode array detection. The validated method is accurate, precise and complies with the acceptance criteria required in the method validation guidelines. Hence, the method will serve as a reliable tool to evaluate the amino acid composition in rice and other cereal matrices to evaluate its nutritional value in terms of protein quality and for future investigation on breeding related to improvement of protein quality of cereal crops.
Conflict of Interest Statement
We wish to confirm that there are no known conflicts of interest associated with this publication and there has been no significant financial support for this work that could have influenced its outcome.
Acknowledgements
This study was supported by the Sri Lanka Treasury. The authors gratefully thank the Industrial Technology Institute for the assistance in this research and rice research development centers (RRDCs) at Batalagoda and Bombuwala of Sri Lanka for providing rice samples.

